Examine the bottom graph on page 462 showing solubility in water as a function of temperature. What
Question:
Examine the bottom graph on page 462 showing solubility in water as a function of temperature. What is the trend for most of the ionic substances shown?
Data from Graph page 462
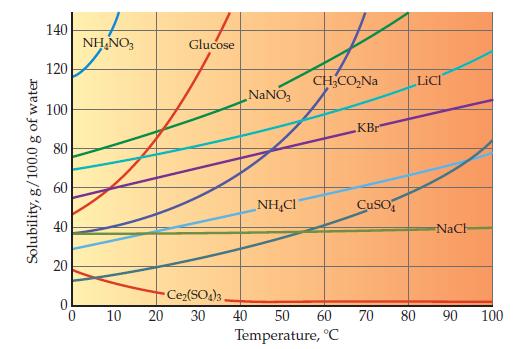
Transcribed Image Text:
Solubility, g/100.0 g of water 140 120 100 80 60 40 20 NH₂NO3 10 Glucose NaNO3 NHẠC CH₂CO₂Na Ce₂(SO4)3, 20 30 40 50 60 Temperature, °C KBr CuSO LICI NaCh 70 80 90 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The trend for most of the ionic substances shown in the bottom graph on page 462 is that their solub...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
One ionic compound in the bottom graph on page 462 shows almost no temperature dependence, and one clearly violates the general trend. Identify these two ionic compounds. Data from Graph page 462...
-
Use the bottom graph on page 462 to determine what mass of water is required to dissolve 50.0 g of NaNO 3 at 40.0 C. Graph from Page 462 Solubility, g/100.0 g of water 140 120 100 80 60 40 20 0 NHNO3...
-
A short-term study was conducted to investigate the effect of mean monthly daily temperature x1 and cost per kilowatt-hour x2 on the mean daily consumption of electricity (in kilowatt-hours, kWh) per...
-
A city levies property taxes of $2 billion in June 2015for its Fiscal year beginning July 1, 2015. The taxes are due by January 31, 2016. The following (in millions) indicates actual and anticipated...
-
JOURNAL ENTRIES FOR MATERIAL, LABOR, AND OVERHEAD Shiar Manufacturing had the following transactions during the month: (a) Purchased raw materials on account, $22,500. (b) Issued direct materials to...
-
What were the benefits of implementing an EDW at Isle? Can you think of other potential benefits that were not listed in the case?
-
How does make determine whether a target needs to be rebuilt?
-
Vulcan Flyovers offers scenic overflights of Mount St. Helens, the volcano in Washington State that explosively erupted in 1982. Data concerning the companys operations in July appear below: The...
-
The force exerted by a 2.4-m massless string on a 0.84-kg object being swung in a horizontal circle is 4.2 N. What is the tangential velocity of the object?
-
Aquatic life is often damaged when hot water is discharged from power stations into rivers and lakes. What might this have to do with gas solubility in water?
-
How is the medical condition known as the bends related to solubility?
-
Dan owns 500 shares of Rocket Corporation common stock. The stock was acquired two years ago for $30 per share. On October 2, 2015, Dan writes five calls on the stock which represents options to buy...
-
A plane pendulum (length I and mass m), restrained by a linear spring with the stiffness k and a linear dashpot of damper with the damping c (see figure below). The upper end of the rigid massless...
-
Dr. Pat has practiced as a Medical Doctor for the past 25 years and his current income is $350,000 per year. Over this time period he left his financial decisions to his wife. She invested his...
-
Find the given value. f(x) = 1 x, f"(-) = 2 F"(-) 2
-
A sample of a gas has a volume of 1000.0 mL at -23.0C and 570. mm pressure. What is the volume of the gas at STP? A. 1,460 mL B. 687 mL C. 751 mL D. 819 mL E. 51.9 mL
-
Write a Python program that calls a user defined function that returns and prints divisors of all numbers between 16 to 18
-
Determine the mode for the followingnumbers. 6. 2 4 3 3 5 4,
-
DC has unused FTC carryover from 2017 in the separate category for GC income as the result of income generated by a foreign branch. The income was foreign source general category income. In 2018 the...
-
Use tabulated standard half-cell potentials to calculate the standard cell potential for the reaction in an electrochemical cell at 25 C: Zn 2+ (aq) + H 2 O 2 (aq) Zn (s) + O 2 (g) + 2 H + (aq)
-
Use the tabulated half-cell potentials to calculate K for the oxidation of nickel by chlorine: Cl 2 (g) + Ni (s) 2 Cl (aq) + Ni 2+ (aq)
-
An electrochemical cell is based on two half-reactions: Oxidation: Fe(s) Fe 2+ (aq, 0.010 M) + 2 e Reduction: Br 2 (l) + 2 e 2 Br (aq, 1.0 M) Compute the cell potential.
-
Is there suffiecent demand to open a logistics firm in london ontario the question is "Detailed findings and management implications for each type of analysis (category, consumer, competitor and...
-
Write an essay on Consumer Behavior in the Age of E-Commerce: Investigate how the rise of e-commerce platforms has altered consumer behavior and decision-making processes. Discuss the impact of...
-
DISCUSSION : 1. Target Markets of Giizghigat Maple Products. 2. Promotion Strategy of of Giizghigat Maple Products. 3. Explain 2 ways to assess the effectiveness of your promotional plan. PLEASE...

Study smarter with the SolutionInn App


