Fluorouracil is a compound administered to cancer patients as a part of chemotherapy. Assign an oxidation state
Question:
Fluorouracil is a compound administered to cancer patients as a part of chemotherapy. Assign an oxidation state to every atom:
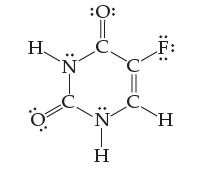
Transcribed Image Text:
H 0 -Z: N :Z-H :: H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The following is the oxidation state assignment for each atom in the molecule fluorouracilC4H3FN2O2 ...View the full answer

Answered By

Rishabh Ojha
During my undergraduate i used to participate as TA (Teaching Assistant) in several electronics and computers subject. I'm passionate about learning Computer Science as my bachelors are in Electronics but i learnt most of the Computer Science subjects on my own which Machine Learning also. At Present, i'm a working professional pursuing my career as a Machine Learning Engineer and i want to help others learn during my free hours, that's all the motivation behind giving tuition. To be frank i have no prior experience of tutoring but i have solved problems on opensource platforms like StackOverflow and github. ~Thanks
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
In methane, CH 4 , (a) Assign an oxidation state to each atom in the molecule. (b) How many electrons does the C atom own by oxidation-state electron bookkeeping? (c) How many more or fewer valence...
-
Consider the organic compound C 3 H 6 O 2 , methyl acetate. (a) What is the problem with using the shortcut method to assign oxidation numbers to the atoms in methyl acetate? (b) The dot diagram of...
-
In chloroform, CHCl 3 , (a) Assign an oxidation state to each atom in the molecule. (b) How many electrons does the C atom own by oxidation-state electron bookkeeping? (c) How many more or fewer...
-
Given the following set of slope staking notes: C Sta 51+00 50+00 49+00 L CX 33.4 F 9.1 33.6. F 10.3 35.4 C 5.5 0.0 F 3.5 R C 3.2 X C 4.1 30.2 0.0 20.0 Bases Base for cut=48 ft Base for fill= 40 ft s...
-
In a recent annual report, eBay Inc. reported that during the year it issued stock of $128 million for acquisitions. How would this be reported on the statement of cash flows?
-
Identify the errors in the following schedule of cost of merchandise sold for the current year ended July 31,2010: Cost of merchandise sold: Merchandise inventory, July 31, 2010 Purchases Plus:...
-
The molecules in the rods and cones in the eye are tuned to absorb photons of particular energies. The retinal molecule, like many molecules, is a long chain. Electrons can freely move along one...
-
Smyth Company is acquired by Radar Corporation on July 1, 2011. Radar exchanges 60,000 shares of its $5 par stock, with a fair value of $20 per share, for the net assets of Smyth Company. Radar...
-
How can you justify that unemployment may occur if the country raises its interest rates? Explain.
-
Assign an oxidation state to each carbon in: (a) H 3 CCH 3 (b) H 2 CCH 2 (c) HCCH
-
Tin cans are actually iron plated with tin. What is the advantage of the tin coating?
-
Refer to the front matter at the beginning of this text and then identify some new features in the 2016 version of Excel.
-
What are the most important things you have learned in this class? What challenged you the most? How will you apply the concepts learned in this course to both your academic and professional life?
-
Begin your initial post by introducing yourself to your classmates. Briefly share your thoughts on why you joined the class and what your short- and long-term goals are regarding financial...
-
A loan arrangement in which a parent company reduces its political risk by using an intermediary bank rather than a direct transfer of funds to a subsidiary is called a(n) Multiple Choice parallel...
-
Pop Eye Company manufactures two types of sailboats, Simple and Complex. Simple is a high-volume product that is relatively simplistic in nature while complex is a low-volume complex manufacturing...
-
Profit, not-for-profit/nonprofit, give a specific example of each type of organization (a business you have heard of, done business with or worked for). For each one, discuss what you foresee as...
-
Rory earns $60,000 per year as a college professor. Latesia is a marketing executive with a salary of $120,000. With respect to the Social Security tax, what are Rory's and Latesia's a. Total taxes?...
-
Find the cross product a x b and verify that it is orthogonal to both a and b. a = (t, 1, 1/t), b = (t 2 , t 2 , 1)
-
For what point in a pumping system is the NPSH computed? Why?
-
It is desired to operate a pump at 1750 rpm by driving it with a four-pole electric motor. For each of the following conditions, compute the specific speed using Eq. (1317). Then, recommend whether...
-
Compute the specific speed for a pump operating at 3500 rpm delivering 500 gal/min of water at a total head of 100 ft. Compare the result with that of Problem 13.40 and with Fig. 13.52. Flow (m3/h)...
-
Following are data from the statements of two companies selling similar products: Current Year-End Balance Sheets Snow Company Zambroni Company Cash Notes receivable-short-term Accounts receivable,...
-
An analysis of the accounts of Teal Mountain Company reveals the following manufacturing cost data for the month ended June 30, 2025. Inventory Beginning Ending Raw materials $9,000 $13,180 Work in...
-
Teradene Corporation purchased land as a factory site and contracted with Maxtor Construction to construct a factory. Teradene made the following expenditures related to the acquisition of the land,...

Study smarter with the SolutionInn App


