Phospholipids are naturally occurring soap like molecules present in the membranes of living cells. A typical phospholipid
Question:
Phospholipids are naturally occurring soap like molecules present in the membranes of living cells. A typical phospholipid structure is:
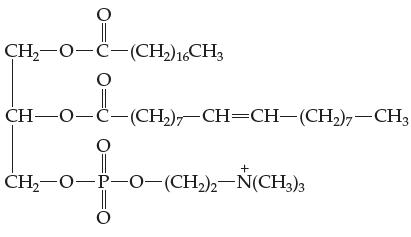
Identify the hydrophobic and hydrophilic portion(s) of this molecule.
Transcribed Image Text:
CH₂-O-C-(CH2) 16CH3 O CH–O-C–(CH,), CH=CH–(CH)–CH3 O CH,—0-P-O–(CH2)2–N(CH3)3 Ö
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
The hydrophobic portion of a phospholipid molecule is the two fatty acid chains These chains are mad...View the full answer

Answered By

Akshay Agarwal
I am a Post-Graduate with a specialization in Finance. I have been working in the Consulting industry for the past 8 years with a focus on the Corporate and Investment Banking domain. Additionally, I have been involved in supporting student across the globe in their academic assignments and always strive to provide high quality support in a timely manner. My notable achievements in the academic field includes serving more than 10,000 clients across geographies on various courses including Accountancy, Finance, Management among other subjects. I always strive to serve my clients in the best possible way ensuring high quality and well explained solutions, which ensures high grades for the students along-with ensuring complete understanding of the subject matter for them. Further, I also believe in making myself available to the students for any follow-ups and ensures complete support and cooperation throughout the project cycle. My passion in the academic field coupled with my educational qualification and industry experience has proved to be instrumental in my success and has helped me stand out of the rest. Looking forward to have a fruitful experience and a cordial working relationship.
5.00+
179+ Reviews
294+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Steve and Alicia were married in a community property state in 1994. The couple's first child, Alexa, was born in 1999, at which time Alicia left her position as a Maricopa County attorney. Alicia...
-
i. In your own words, provide a description of what happened in the case. Your description should include more than one source and detail what happened to someone who does not know anything about the...
-
Locate your job description at your workplace If you do not have a job description go online to Monster com and search on the Jobs tab at the top Review a job description of your choice then compare...
-
In the light of what you know about IHRM, how easy do you think it is to manage a global employer brand? What issues would an IHRM professional expect to have to manage? To answer this, analyse the...
-
Sam Santiago operates a retail variety store. The books include a general journal and an accounts payable ledger. Selected account balances on May 1 are as follows: General Ledger Cash .............
-
The postmaster of a small western town receives a certain number of complaints each day about mail delivery. Determine three- sigma control limits using the following data. Is the process incontrol?...
-
Mind & Motion entered into a real estate purchase contract (REPC) with Celtic Bank to buy a large piece of the banks foreclosed property. Although the county had approved plans to construct...
-
Use this information to prepare a net worth analysis of Slicks girlfriend. Steve (Slick) Willy, 45, just got out of jail. As a reformed citizen on parole, Slick decides to go into business for...
-
Why is it important to learn SQL? In which jobs you need to know SQL? 2A- What is the definition of DML and DDL? Give examples in each case. 2B- Consider the following two tables below: VENDOR and...
-
Why do most solids become more soluble in water with increasing temperature?
-
What is the mass in grams of glucose dissolved in 60.0 g of water at 20.0C if the solution is saturated?
-
If a line is horizontal, then its slope is _______.
-
If the 10-year Treasury bond rate is 9.0%, the inflation premium is 2.0% and maturity risk premium on 10-year bond is .3%: Why would an investor purchase a treasury bond, versus a technical corporate...
-
Students should replace the ## with the last two digits of their student number. The measurements are in feet. Use the last two digits of your student # as benchmark elevation. BS (+S) FS (-S) BM 1...
-
Samantha purchased personal property for her business for $635,000 on January 5, 2023. What amount can she deduct by using bonus depreciation?
-
Describe the concept of cost behaviour. Explain why it is important to understand cost behaviour? Orient Trading Co. sells products B and C. Sales for February were 400 units of B at $25 and 800...
-
2. martin corporation's common stock sells for $55 per share. the current dividend is $2.75 per share; dividends are expected to grow at 5.00 percent per year indefinitely. the floatation cost of new...
-
A telephone survey conducted by the Maritz Marketing Research company found that 43% of Americans expect to save more money next year than they saved last year. Forty-five percent of those surveyed...
-
Carlton Stokes owns and operates a car-detailing business named SuperShine & Detailing. For $150, Carltons business will hand wash and wax customers cars, vacuum the interior, and thoroughly clean...
-
The composite beam is constructed from wood and reinforced with a steel strap. Use the method of Sec. 6.6 and calculate the maximum shear stress in the beam when it is subjected to a shear of V = 50...
-
The beam has a rectangular cross section and is subjected to a load P that is just large enough to develop a fully plastic moment M p = PL at the fixed support. If the material is elastic perfectly...
-
The beam in Fig. 648f is subjected to a fully plastic moment M p . Prove that the longitudinal and transverse shear stresses in the beam are zero. Hint: Consider an element of the beam shown in Fig....
-
Give the prime-power factorization of each of these numbers. (a) 15 (b) 3375 (a) The prime-power factorization of 15 is (c) 37,000
-
Geyser Manufacturing replaced its hourly quality inspectors with a computerized inspection system. What will be the impact of this choice on the company's degree of operating leverage?
-
2 . Husband ( H ) and Wife ( W ) are both lawyers and practice law together. In each of the following alternatives, please determine if they are entitled to QBD for 2 0 2 2 and, if so , in what...

Study smarter with the SolutionInn App


