Some liquid water is placed in a room-temperature container and sealed. After some time, the level of
Question:
Some liquid water is placed in a room-temperature container and sealed. After some time, the level of the liquid drops by a small amount. After that, the water level remains constant.
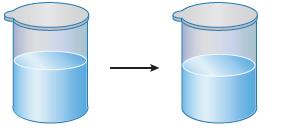
What does this have to do with equilibrium?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
This scenario relates directly to the concept of equilibrium Heres the breakdown Initial State Water ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
A container filled with 45 kg of liquid water at 95°C is placed in a 90-m3 room that is initially at 12°C. Thermal equilibrium is established after a while as a result of heat transfer...
-
A container filled with 45 kg of liquid water at 95°C is placed in a 90-m3 room that is initially at 12°C. Thermal equilibrium is established after a while as a result of heat transfer...
-
Some water is placed in a sealed glass container connected to a vacuum pump (a device used to pump gases from a container), and the pump is turned on. The water appears to boil and then freezes....
-
Using atomic weight, crystal structure, and atomic radius data tabulated inside the front cover of the book, compute the theoretical densities of aluminum (Al), nickel (Ni), magnesium (Mg), and...
-
Analyzing and Interpreting Return on Assets Tiffany & Co. is one of the worlds premier jewelers and a designer of other fine gifts and housewares. Presented here are selected income statement and...
-
A cereal company wants to see which of two promotional strategies, supplying coupons in a local newspaper or including coupons in the cereal package itself, is more effective. The company randomly...
-
On October 1, 2017, Gordon borrows \($150\),000 cash from a bank by signing a three-year installment note bearing 10% interest. The note requires equal payments of \($60\),316 each year on September...
-
After watching the video on expatriates, discuss the following: 1. What kind of staffing approach is being used by Dubai? Explain. 2. What does Kevin ONeal, as an expatriate, bring to Dubai? 3. What...
-
As an International Business Strategist advise on how Porter's Diamond is a useful concept in explaining home and host location strategies in International Business? Your answer should make reference...
-
Consider the reversible reaction shown below: Instead of using molar concentrations, you can use gas pressures to calculate an equilibrium constant, in which case it is often labeled K p instead of K...
-
A sealed 1-L flask is filled with 1 mole of H 2 (g) and 1 mole of I 2 (g). The reversible reaction H 2 (g) + I 2 (g) 2HI(g) ensues. A plot showing what happens to the HI concentration as a percent...
-
Describe the six value chain business activities that this product would pass through from its inception to its ultimate delivery to the customer.
-
Zeta Private Corp has just paid a dividend of $4.54 per share. The company's current stock price is $44.42 each. If the dividends are expected to grow at a constant rate of 5.2% per year. Determine...
-
Carrot the cat likes taking naps in one of four locations: the rug, the bed, the ledge, and the sink. Carrot has the following conditions: He will not sleep in the sink twice in a row He will sleep...
-
Based on the Bursa Malaysia's Listing Requirements, discuss the obligation of a listed issuer to comply on matters of related party transaction and also discuss and explain Audit Committee Review ,...
-
You are evaluating a project with an initial investment of $9.6 million, and equal expected cash flows of $5.1 million per year for years 1 to 5. What is this projects simple payback? The corporate...
-
What do you think about carbon offsets as a method to reduce greenhouse effects?
-
Below is the income statement and balance sheet of Closely Held Corporation. From this information, prepare a statement of cash flows for the year ended September 30, 2005 2004 2005 (Decrease) Assets...
-
(a) With not more than 300 words, examine why Malaysia missed its 2020 targets to divert 40% of waste from landfills and increase recycling rates to 22%. (10 marks) b) Why arent Malaysian recycling?...
-
In SI units, acceleration is measured in units of meters per second squared (m/s 2 ). Which of the following combinations of units can also be used to measure acceleration? (a) Cm/s (b) Cm/s 2 (c) m...
-
Consider the motion of a sprinter running a 100-m dash. When it is run outdoors, this race is run along a straight-line portion of a track, so it is an example of motion in one dimension. Draw...
-
A car is initially at rest on an icy road that is extremely slippery. The driver finds that he is unable to get the car to drive away because the wheels simply spin when he tries to accelerate....
-
Solve the following optimization problems. (a) min z = f(x) = x+3x-4 subject to x = R. (b) min = f(x) = xe subject to x = R.
-
Summarize this paragraph to fit into 2-3 slides for a power-point presentation. Only take out important key elements. Two potential pricing strategy options for a B2B market could be cost-plus...
-
A corporation considering expanding into overseas markets needs to be aware of a variety of dangers. The first factor is the country's political and economic stability, both of which are crucial for...

Study smarter with the SolutionInn App


