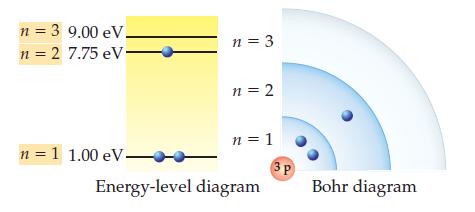
The ground state for the lithium (Li) atom and the scaled energies of its shells are shown
Question:
The ground state for the lithium (Li) atom and the scaled energies of its shells are shown below. Draw a Bohr diagram for the lowest-energy excited state of lithium.
Transcribed Image Text:
n = 3 9.00 eV. n = 2 7.75 eV. n = 1 1.00 eV- n = 3 n = 2 n = 1 Energy-level diagram 3P Bohr diagram
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Two excited states are drawn below but only a correctly an...View the full answer

Answered By

Loise Ndungu
I have five years of experience as a writer. As I embark on writing your papers from the prologue to the epilogue, my enthusiasm is driven by the importance of producing a quality product. I put premium product delivery as my top priority, as this is what my clients are seeking and what makes me different from other writers. My goal is to craft a masterpiece each time I embark on a freelance work task! I'm a freelance writer who provides his customers with outstanding and remarkable custom writings on various subjects. Let's work together for perfect grades.
4.90+
78+ Reviews
224+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Draw a Bohr diagram for the highest-energy excited state you can make using the three shells shown in Practice Problem 4.9 and exciting only a single electron. Data from Problem 4.9 The ground state...
-
A hydrogen atom is excited from its ground state to the state with n = 4. (a) How much energy must be absorbed by the atom? Consider the photon energies that can be emitted by the atom as it...
-
Draw all of the p molecular orbitals for (3E)-1,3,5-hexatriene, order them from lowest to highest in energy, and indicate the number of electrons that would be found in each in the ground state for...
-
Refer to the facts presented in problem P7-13. In problem On January 25, 2011, Douglas Ltd. purchased 1,000 common shares of BMO (Bank of Montreal) for $65 each. During the remainder of 2011, Douglas...
-
Compute the equivalent production (unit output) for the month for each of the followingsituations: Units Completed During Month 10,000 22,000 8,000 Units in Process, End of Month 5,000 4,000 1,000...
-
On December 31, 20X3, Broadway Corporation reported common stock outstanding of $200,000, additional paid-in capital of $300,000, and retained earnings of $100,000. On January 1, 20X4, Johe Company...
-
Nereus Montemayor was an employee of VZ Hogs, a company that raises hogs and produces hog feed. VZ Hogs used an extruder manufactured by Sebright Products, Inc. to create hog feed out of discarded...
-
Comm Devices (CD) is a division of Worldwide Communications, Inc. CD produces pagers and other personal communication devices. These devices are sold to other Worldwide divisions, as well as to other...
-
Relational Databases are based on relational models and relational math. Write a brief description and history of the relational model.?
-
Ionization energy increases left to right across a period because (a) It does not. Ionization energy decreases left to right across a period. (b) As you go down a group, atoms increase in size due to...
-
Locate the elements in group VIIA in the ionization energy plot. Do any of these elements violate the expected trend in ionization energies?
-
Capri Classic Inc. has the following balances in its shareholders' equity accounts as at October 31, 2015: Retained earnings ..............................................$2,561,000 Common shares...
-
Gasworks AG has been approached to sell up to 5 million litres of gasoline in 3 months at a price of 1.85 per litre. Gasoline is currently selling on the wholesale market at 1.65 per litre and has a...
-
Efficient Markets Hypothesis When the 56-year-old founder of the Turkish firm, Gulf Oil and Minerals, died of a heart attack, the share price immediately jumped from 18.00 Lira a share to 20.25 Lira,...
-
(a) Many explanations and justifications are made by acquiring (and sometimes target) managers for a merger. Review these justifications and discuss whether they are good or bad for shareholders. (b)...
-
Efficient Markets Hypothesis Your broker commented that well-managed firms are better investments than poorly managed firms. As evidence your broker cited a recent study examining 100 small...
-
Eurocargoair is a British private air courier firm that has been given the option to purchase three new small jets at the price of 3 million per plane. The purchase agreement is only valid for the...
-
PIM Industries, Inc., manufactures electronics components. Each unit costs $30 before the final test. The final test rejects, on average, 5 percent of the 50,000 units manufactured per year. The...
-
The May 2014 revenue and cost information for Houston Outfitters, Inc. follow: Sales Revenue (at standard).............. $ 540,000 Cost of Goods Sold (at standard) ..........341,000 Direct Materials...
-
The bond angle about oxygen in alcohols and ethers is typically quite close to tetrahedral (109.5°), but opens up significantly in response to extreme steric crowding; for example, in going from...
-
Each of the carbons in ethane is surrounded by four atoms in a roughly tetrahedral geometry; each carbon in ethene is surrounded by three atoms in a trigonal planar geometry and each carbon in...
-
VSEPR (valence state electron pair repulsion) theory was formulated to anticipate the local geometry about an atom in a molecule (see discussion in Section 25.1). All that is required is the number...
-
give explanations and answers for all each point Text File Process . Pipe and redirection First n lines Grep lines with word "apple" Grep lines with word1 or word2 Output number of lines with word...
-
Cameron takes a road trip. He starts from his home in the suburbs of Cleveland, OH, and travels to Pittsburgh, PA, to visit his aunt and uncle. The distance Cameron drives from Cleveland to...
-
How to compute the Net Profit Margin in San Miguel Corporation's 2021 Annual Report?

Study smarter with the SolutionInn App


