Which molecule is more unsaturated and why?: (a) (b)
Question:
Which molecule is more unsaturated and why?:
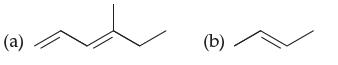
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Explanation a E4methylhexa13diene b Ebut2ene a Moleculea is more unsaturated than molecule ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Suppose you are asked to make molecule (a) of Problem 17.44 even more unsaturated by removing H atoms to form a third double bond. (a) Why cant this be the molecule you form? (b) Show a line drawing...
-
Write a paper on "IMPACT OF USAID ON NEPAL'S SOCIO-ECONOMIC DEVELOPMENT" and the question is "TO WHAT EXTENT HAS THE USAID CONTRIBUTED TO STATE SECURITY IN NEPAL POST EARTHQUAKE?
-
Draw all possible line drawings for the unsaturated hydrocarbon molecule in Practice Problem 17.5. Data from Practice Problem 17.5 A particular linear hydrocarbon molecule has six carbons and ten...
-
i don?t understand why in a), NaOCH3 is strong base and strong nucleophile. In d) CH3COONa is weak base and weak nucleophile. In e) NaSCH2CH3 is weak base and strong nucleophile. Is there any trick...
-
Upscale hotels in the United States recently cut their prices by 20 percent in an effort to bolster dwindling occupancy rates among business travelers. A survey performed by a major research...
-
How does a shelf registration differ from other public security offerings?
-
Assume Evco, Inc., has a current price of \($45\) and will pay a \($2.05\) dividend in one year, and its equity cost of capital is 16%. What price must you expect it to sell for right after paying...
-
Jan Gentry is the owner of a small company that produces electric scissors used to cut fabric. The annual demand is for 8,000 scissors, and Jan produces the scissors in batches. On the average, Jan...
-
Recall a work or personal situation in which a particular individual tried to get you or a group to make a change, then respond to the questions below: Provide a little background so that we...
-
A molecule contains 5 carbon atoms and 12 hydrogen atoms. Is it saturated or unsaturated? Explain how you know.
-
Which molecule in Problem 17.39 is unsaturated? Data from Problem 17.39 Convert each line drawing to a structural formula, then give the molecular formula and describe the hydrocarbon as being linear...
-
Use the information in P 23-3 to prepare a chargedischarge statement for the estate of George Wilson for the period March 1, 2011 through April 30, 2011.
-
In what two specific ways can market prices be used as a measure of resource scarcity? From your reading of Chapter 1, are you able to sense why market prices may "fail" to measure the scarcity of...
-
In the blank next to each of the following terms, place the letter A through D that corresponds to the description of that term. Some letters are used more than once. 1. Activity-based costing 2....
-
1.)Apple reports in notes to its financial statements that, in addition to its products sold, it includes the following costs (among others) in cost of sales: customer shipping and handling expenses...
-
Logicalize the following, if necessary: Type and copy invoice Collate customer details SR1 form blue File details from new customer View patients name and address Photocopy application form ...
-
Identify whether each description most likely applies to managerial (M) or financial (F) accounting. 1. Its primary users are company managers. 2. Its information is often available only after an...
-
How could we implement process costing in a plant that has a sequential production process? For example, assume a plant has two separate departmentsparts are fabricated in department 1 and assembled...
-
How has the too-big-to-fail policy been limited in the FDICIA legislation? How might limiting the too-big-to-fail policy help reduce the risk of a future banking crisis?
-
With the previous problem in mind, return to the autocorrelation of a sine function, shown in Fig. 11.51. Now suppose we have a signal composed of a great many sinusoidal components. Imagine that you...
-
Imagine that we have the arrangement depicted in Fig. 12.8. If the separation between fringes (max. to max.) is 1 mm and if the projected width of the source slit on the screen is 0.5 mm, compute the...
-
Referring to the slit source and pinhole screen arrangement of Fig. P.12.13, show by integration over the source that Fig. P.12.13 sin (/Al)b cos (2/As) I() b + /Al S2 S' S1
-
how do I record a check drawn for $ 8 8 0 that has been in correctly charged by the bank as $ 8 0 on a journal entry?
-
In R programming use Laplace approximation to find mean and standard deviation. data = rexp(20, rate=0.5) . Suppose you model this data as following an Exponential( p ) distribution, where the prior...
-
Y ltd purchased a debt security for sh . 5 m at a discount for sh . 4 , 6 7 0 , 0 0 0 on 1 st Jan, 2 0 0 4 . The debt is repayable in 5 yrs time at an interest rate of 6 % p . a payable annually in...

Study smarter with the SolutionInn App


