Which of the following reactions is spontaneous? 2+ 3 Zn+ + 2 Al-3 Zn + 2 A1+
Question:
Which of the following reactions is spontaneous?
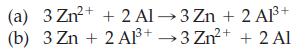
Transcribed Image Text:
2+ 3 Zn²+ + 2 Al-3 Zn + 2 A1³+ (a) (b) 3 Zn + 2 2+ Al³+3 Zn²+ + 2 Al
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
3 Zn2 2 Al 3 Zn 2 Al3 This reaction is spontaneous We can determine this by using standard electr...View the full answer

Answered By

Collins Omondi
I have been an academic and content writer for at least 6 years, working on different academic fields including accounting, political science, technology, law, and nursing in addition to those earlier listed under my education background.
I have a Bachelor’s degree in Commerce (Accounting option), and vast knowledge in various academic fields Finance, Economics, Marketing, Management, Social Science, Women and Gender, Business law, and Statistics among others.
4.80+
4+ Reviews
16+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
From the values of H and S, predict which of the following reactions would be spontaneous at 25C: Reaction A: H 5 10.5 kJ/mol, DS 5 30 J/K ? mol; reaction B: H 5 1.8 kJ/mol, S 5 2113 J/K ? mol. If...
-
According to Scientist 2s viewpoint, which of the following observations provides the strongest argument against using placebos in clinical research? A. The fact that patients sometimes deceive...
-
(A) Which of the four cases in Table 13.3 would apply to each of the following reactions? (B) Under what temperature conditions would the following reactions occur spontaneously? (a) The...
-
Although illegal, overloading is common in the trucking industry. A state highway planning agency (Minnesota Department of Transportation) monitored the movements of overweight trucks on an...
-
Milken Manufacturing has three product lines. The companys new accountant, Marvin LaSance, is responsible for allocating facility-level costs to these product lines. Mr. LaSance is finding the...
-
Gonzalez Corporation was authorized to issue $100,000 of 7%, four-year bonds, dated May 1, 2012. All the bonds were sold on that date when the effective interest rate was 8%. Interest is payable on...
-
The circuit shown in Figure Q26.28 has a resonance frequency of \(15 \mathrm{kHz}\). What is the value of \(L\) ? A. \(1.6 \mu \mathrm{H}\) B. \(2.4 \mu \mathrm{H}\) C. \(5.2 \mu \mathrm{H}\) D. \(18...
-
Brenda and Sasha are equal partners in an interior decorating business in Kenora, Ontario. Their income statement for the year ended December 31 shows: They have each asked you to prepare their...
-
5. Oil is leaking from a pipeline on the surface of Laguna Lake and forms an oil slick whose volume increases at a constant rate of 2000 cm/min. The oil slick takes the form of a right circular...
-
Using a diagram, show how you would construct a zincaluminum battery. Label the cathode, the anode, the + electrode, and the electrode. Indicate the direction of electron flow, and identify the...
-
What will happen if a piece of Zn metal is placed in a solution of Al 3+ ions?
-
Recall that in a standard deck of 52 cards there are 12 picture cards - four each of jacks, queens, and kings. Kevin draws one card from the deck. Find the probability his card is a king if we know...
-
Nonverbal Communication: Analyzing Nonverbal Signals Write a list of the categories of nonverbal communications identified in this chapter. Take this list, a notebook, and a pen into a public...
-
How do you think different ethnocentric approaches to doing business might damage international trade?
-
You have been invited to speak at an annual industry conference. After preparing the outline for your presentation, you see that youve identified 14 different points to support your main idea. Should...
-
How might team norms encourage groupthink?
-
You manage your companys software development lab, which has added several dozen new employees over the past year. One of the senior developers runs into you at lunch and starts to unload his...
-
For years, courts, employers, and employees have struggled with the issue of employee drug testing. Employers want their employees to be drug free, whereas employees do not want to submit to an...
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
Determine the optical path difference for the two waves A and B, both having vacuum wavelengths of 500 nm, depicted in Fig. P.7.6; the glass (n = 1.52) tank is filled with water (n = 1.33). If the...
-
Using Eqs. (7.9), (7.10), and (7.11), show that the resultant of the two waves and is = E01 sin [wt k(x + Ax)] E1 %3D E2 = E01 sin (wt kx)
-
Add the two waves of Problem 7.7 directly to find Eq. (7.17). Data from Problem 7.7 Using Eqs. (7.9), (7.10), and (7.11), show that the resultant of the two waves and is k r x E = 2E01 cos (7.17) sin...
-
1. Describe mission, vision and culture. 2. Explain 3 reasons why a start-up organization would have a written business plan. 3. What is the importance of having a professional, polished business...
-
Consider the region, R, bounded by y=x+1 and y=x+1 in the first quadrant. a. Set up the integral needed to find the volume of the solid generated by revolving R around the line x=2 with the indicated...
-
Integrity is a word used and misused often. Find an example of someone with, or someone without integrity that are public business figures. Post the name and explain what it was they did or did not...

Study smarter with the SolutionInn App


