Which of the following represent a chemical transformation? (a) 4 P(s) + 5O(g) 2 PO5(s) (b)
Question:
Which of the following represent a chemical transformation?
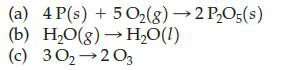
Transcribed Image Text:
(a) 4 P(s) + 5O₂(g) → 2 P₂O5(s) (b) H₂O(g) → H₂O(1) (c) 30₂ 203
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a and c because the produc...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Atoms First
ISBN: 9780321927118
5th Edition
Authors: Steve Russo And Michael Silver
Question Posted:
Students also viewed these Sciences questions
-
Determine which of the following represent potential energy and which represent kinetic energy. (a). Thermal energy (b). Gravitational energy (c). Chemical energy (d). Electrostatic energy
-
Which of the following represent configurations of thallium ions in compounds? Explain your decision in each case. a. Tl2+ [Xe]4f145d106p1 b. Tl3+ [Xe]4f145d10 c. Tl4+ [Xe]4f145d9 d. Tl+...
-
Which of the following structures represent the same compound? Which ones represent different compounds? (a) (b) (c) (d) (e) (f) (g) Name the structures given in Problem 3-33, parts (a), (c), (e),...
-
Sugar (C12H22O11) is a molecular compound that stays together inwater, while NaCl and MgSO4?7H2O are ionic compounds that dissociate into cations andanions as illustrated in the NaCl example below:...
-
Locate the 2007 financial statements for The Walt Disney Company on the Internet. Use those financial statements and consider the following questions. 1. As illustrated in Exhibit 10-10, Interbrand...
-
What are the values of the engineering economy symbols P, F, A, i, and n in the following functions? Use a question mark for the symbol that is to be determined. (a) NPER (8%, 1500,8000,2000) (b) FV...
-
Many single women and married couples use donated sperm to conceive children each year. Pennsylvania resident Donna Donovan decided to use donated sperm from Idant Laboratories, a New York sperm bank...
-
Accounting for Research and Development Costs Czeslaw Corporations research and development department has an idea for a project it believes will culminate in a new product that would be very...
-
How does a Cloud Data Management Platform differ from a Database Management System?
-
Water (H 2 O) and carbon dioxide (CO 2 ) are produced in a chemical reaction when methane (CH 4 ) and oxygen (O 2 ) are combined and heated. Which are the reactants, and which are the products?
-
You are presented with a block made of some pure metal and told the metal is gold, but you have your doubts. Using a thermometer, how can you determine whether the metal is gold?
-
The production department described in Exercise 20-8 had $531,480 of direct materials and $407,689 of direct labor cost charged to it during April. Also, its beginning inventory included $74,075 of...
-
Health System experienced a 10% net increase in cash flow in 2021. While cash flow increased, the net income declined by 10%. In your own words, what are some reasons this could have occurred? Hint:...
-
Which taxpayer has propertyFor Tax Year 2022, a taxpayer generally may elect to immediately claim up to what amount for the Section 179 deduction? that is depreciable? Kelly owns a townhouse and...
-
Discuss whether capital markets are efficient or not, and how public good news like firm announcing that the actual earnings per share is greater than market expectations would affect the firm's...
-
Using the Percentage of Sales method, you calculated your Cost of Goods Sold to be $5,000, on $20,000 in sales for last month. Using the same method, what will your Cost of Goods Sold be for this...
-
Jeff loaned Mike $8,000, which Mike agreed to repay with interest. Mike is making monthly payments of $350 to Jeff, which includes $50 in interest. How should Jeff report this income? Jim is not...
-
Instar Company has several investments in other companies. The following information regarding these investments is available at December 31, 2010. 1. Instar holds bonds issued by Dorsel Corp. The...
-
1. What are some current issues facing Saudi Arabia? What is the climate for doing business in Saudi Arabia today? 2. Is it legal for Auger's firm to make a payment of $100,000 to help ensure this...
-
How many constitutional isomers are obtained when each of the following compounds undergoes monochlorination? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j)
-
Propylene is produced by cracking petroleum and is a very useful precursor in the production of many useful polymers. Propylene has one constitutional isomer. Draw that isomer, and identify its...
-
Identify the reagents you would use to achieve each of the following transformations: (a) (b) (c) (d) (e) OTs `CN
-
Describe the impact the Common law has on the health care industry Identify the name of a Common law that was enacted to protect confidentiality in the health care industry.
-
Retail Method of Estimating Inventory The records of Earthly Goods provided the following information for the year ended December 2 1 2 0 2 0 At Cost At Retail January 1 beginning inventory $ 9 4 2...
-
Interview 3 content creators/writers. Minimum 5K followers on any Social Media platforms Come up with 5-7 content related questions for them to answer. At least 1 of them has to be Canadian based....

Study smarter with the SolutionInn App


