Balance each of the following chemical equations by inspection. (a) Co(s) + O(g) CoO3(s) (b) LiClO3(s)
Question:
Balance each of the following chemical equations by inspection.
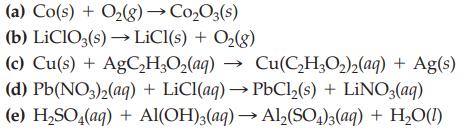
Transcribed Image Text:
(a) Co(s) + O₂(g) → Co₂O3(s) (b) LiClO3(s) → LiCl(s) + O₂(g) (c) Cu(s) + AgC₂H₂O₂(aq) → Cu(C₂H₂O₂)2(aq) + Ag(s) (d) Pb(NO3)2(aq) + LiCl(aq) →PbCl₂(s) + LiNO3(aq) (e) H₂SO4(aq) + Al(OH)3(aq) → Al₂(SO4)3(aq) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Cos Og CoOs This equation is already balanced There are 1 cobalt atom on each side 2 oxygen atoms ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Balance each of the following chemical equations by inspection. (a) PCl 5 (s) + H 2 O(l) H 3 PO 4 (aq) + HCl(aq) (b) TiCl 4 (s) + H 2 O(g) TiO 2 (s) + HCl(g).
-
Balance each of the following chemical equations by inspection. (a) F 2 (g) + NaBr(aq) Br 2 (l) + NaF(aq) (b) Sb 2 S 3 (s) + HCl(aq) SbCl 3 (aq) + H 2 S(g).
-
Balance each of the following chemical equations by inspection. (a) FeO(l) + Al(l) Al 2 O 3 (l) + Fe(l) (b) MnO 2 (l) + Al(l) Al 2 O 3 (l) + Mn(l).
-
You bought a share of 3.4 percent preferred stock for $96.82 last year. The market price for your stock is now $98.34. What is your total return for last year?
-
The cash register tape for Kelly Kapoor Industries reported sales of $6,891.50. Record the journal entry that would be necessary for each of the following situations. (a) Cash to be accounted for...
-
What is a marketing dashboard? How can it be used?
-
Yazaki Corporation and DENSO Corporation are both Japanese companies that supply electrical components to manufacturers of automobiles. Following an investigation by the U.S. Department of Justice in...
-
Eastport Companys operating budgets reveal the following information: net sales, $400,000; beginning materials inventory, $23,000; materials purchased, $185,000; beginning work in process inventory,...
-
22 22 The atomic number of an element T is 10. T shows similar properties to an element with atomic number 23. 24. A. 14 B. 16 C. 18 d. 20 The electron configurations of elements W, X, Y and Z are...
-
Balance each of the following chemical equations by inspection (a) Pb(s) + O(g) PbO(s) (b) LINO3(s)LiNO(s) + O(g) (c) Mg(s) + HCHO(aq) Mg(CHO)2(aq) + H(g) (d) Hg2(NO3)2(aq) + NaBr(aq) HgBr(s) +...
-
Write a balanced chemical equation for each of the following neutralization reactions: (a) Chloric acid neutralizes a strontium hydroxide solution. (b) Phosphoric acid neutralizes a sodium hydroxide...
-
What additional issues are involved with miscellaneous, other, and clearing accounts?
-
To be covered by Philhealth, a member should: a. must have paid at least three months' premium contributions within the immediate six-month period prior to the first day of confinement b must have...
-
The plan is to search the whole record of all rainfall data for a particular station's data, and then present this data on the client screen. This search should be done in a thread. Consider the...
-
In this assignment, you must adopt the inverted pyramid style and prepare a plan for a story about an incident that has just occurred. The incident must be something that has happened within the last...
-
6. Consider a two-layer neural network as shown in Fig. Q6 with ReLU activation functions in the hidden layer and the output layer. The initial weights, biases and training inputs/outputs for a...
-
1. The Corona Virus Disease 2019 (COVID-19) has been declared a worldwide pandemic, and a key method for diagnosing COVID-19 is chest X-ray imaging. Suppose that we have dataset of 448 COVID-19 chest...
-
Use the appropriate function in Excel to calculate the annual straight-line (SL) depreciation charge for an asset that has a $10,000 acquisition cost, a salvage value of $500, and a useful life of...
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
Depreciation for Partial PeriodSL, SYD, and DDB Alladin Company purchased Machine #201 on May 1, 2010. The following information relating to Machine #201 was gathered at the end of May....
-
Depreciation for Partial PeriodsSL, Act, SYD, and DDB the cost of equipment purchased by Charleston, Inc., on June 1, 2010 is $89,000. It is estimated that the machine will have a $5,000 salvage...
-
Depreciation?SYD, Act., SL, and DDB the following data relate to the Plant Assets account of Eshkol, Inc. at December 31, 2010. *In the year an asset is purchased, Eshkol, Inc. does not record any...
-
Use conversion algorithm to convert ER diagram to Relational Model Or All ER to relational model intermediatory conversion steps with final relational model (i.e. step 4) RUberID REmail Passwd...
-
A company is planning to purchase and store two items, gadgets and widgets. Each gadget costs $2.00 and occupies 2 square meters of floor space; each widget costs $3.00 and occu pies 1 square meter...
-
Relational Databases are based on relational models and relational math. Write a brief description and history of the relational model.?

Study smarter with the SolutionInn App


