Based on the general trends in the periodic table, predict which element in each of the following
Question:
Based on the general trends in the periodic table, predict which element in each of the following pairs has the higher ionization energy:
(a) Na or Mg
(b) O or S.
Periodic Table:
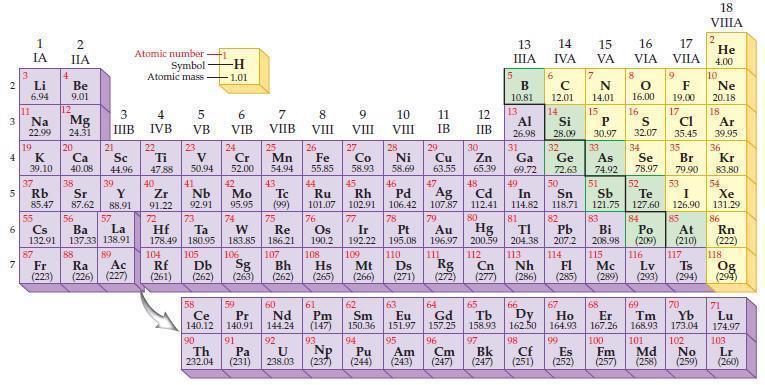
Transcribed Image Text:
2 3 4 15 6 7 3 11 Li 6.94 Na 22.99 19 1 IA 37 R Rb 55 4 87 2 IIA Be 9.01 12 K Ca Sc 39.10 40.08 44.96 Mg 24.31 20 38 21 Sr Y 85.47 87.62 88.91 3 IIIB 88 39 57 56 Cs La Ba 132.91 137.33 138.91 89 Fr Ra Ac (223) (226) (227) Atomic number Symbol Atomic mass 4 IVB 22 Ti 47.88 40 Zr 2 91.22 72 5 VB 104 23 V 50.94 41 73 Hf Ta 178.49 180.95 105 Rf Db (261) (262) -H -1.01 6 VIB Nb Mo 92.91 95.95 90 24 Cr 52.00 42 74 106 Sg (263) 58 Pr Ce 140.12 140.91 91 59 7 VIIB Th Pa 232.04 (231) 25 W Re 183.85 186.21 Mn 54.94 43. Tc (99) 75 107 Bh (262) 60 Nd 144.24 92 U 238.03 8 VIII 26 Fe 55.85 76 Os 190.2 61 Pm (147) 93 NP 9 VIII (237) 10 VIII 11 IB 12 IIB 27 28 29 Zn Co Ni Cu 58.93 58.69 63.55 65.39 44 47 48 49 45 46 Ru Rh Pd Ag Cd In 101.07 102.91 106.42 107.87 112.41 114.82 77 78 80 Ir Pt 192.22 195.08 109 110 Hs Mt Ds Rg Cn (265) (266) (271) (272) (277) 79 Au Hg 196.97 200.59 111 112 108 30 5 13 ΠΙΑ B 10.81 13 6 14 15 IVA с 12.01 14 81 82 TI Pb 204.38 207.2 113 114 Nh Fl (286) (285) 17 16 VA VIA VILA 66 67 62 63 64 65 Sm Eu Gd Tb Dy Ho 150.36 151.97 157.25 158.93 162.50 164.93 99 94 95 96 97 Pu Am Cm Bk (244) (243) (247) (247) 7 N 14.01 15 Al Si 31 34 26.98 28.09 32 33 Ga Ge As Se 69.72 72.63 74.92 78.97 50 51 52 Sn Sb Te I 118.71 121.75 127.60 126.90 53 P 30.97 8 O 16.00 68 16 98 167.26 100 Cf Es Fm (251) (252) (257) 9 F 19.00 17 Cl 35.45 S 32.07 35 83 84 85 Bi Po At 208.98 (209) (210) 115 116 117 Mc Lv Ts (289) (293) (294) Br 79.90 69 70 Er Tm Yb 168.93 173.04 101 102 Md No (258) (259) 18 VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39,95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 Lr (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a...View the full answer

Answered By

Sheikh Muhammad Ibrahim
During the course of my study, I have worked as a private tutor. I have taught Maths and Physics to O'Level and A'Level students, as well as I have also taught basic engineering courses to my juniors in the university. Engineering intrigues me alot because it a world full of ideas. I have passionately taught students and this made me learn alot. Teaching algebra and basic calculus, from the very basics of it made me very patient. Therefore, I know many tricks to make your work easier for you. I believe that every student has a potential to work himself. I am just here to polish your skills. I am a bright student in my university. My juniors are always happy from me because I help in their assignments and they are never late.
4.90+
14+ Reviews
24+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
If $1,000 is put into a savings account at 8% interest compounded continuously, how much will it be worth at the end of 4 years?
-
Based on the general trends in the periodic table, predict which element in each of the following pairs has the higher ionization energy: (a) Li or Na (b) O or F. Periodic Table: 2 3 4 in 6 3 7 Li...
-
Based on the general trends in the periodic table, predict which element in each of the following pairs has the most metallic character: (a) Sn or Pb (b) Ag or Sr (c) Al or B (d) Ga or Ge. Periodic...
-
If possible, completely factor the expressions in Problems 336. 9-x-2x
-
Revenue and Related Transactions. During its current fiscal year, Dearborn General Hospital, a not-for-profit health care organization, had the following revenue-related transactions (amounts...
-
What are the major advantages of the asset and liability method?
-
The following categories appear on the income statement of Joshua Jeans Company: (a) Net sales. (b) Cost of sales. (c) Operating expenses. (d) Other revenue/expense. (e) Income tax expense. Classify...
-
An investor is considering investing in the long-term bonds and common stock of Companies M and N. Both firms operate in the same industry. Both also pay a dividend per share of $8 and have a yield...
-
An investor wants to lock in the interest rate for a three-month period beginning three months from today on a principal of $4 million. To achieve this purpose, the investor enters into a forward...
-
Which group in the periodic table has the highest ionization energy? Which group has the lowest ionization energy? Periodic Table: 2 3 4 10 6 3 7 11 Li 6.94 1 IA Na 22.99 19 37 5 Rb K 39.10 al 55 Cs...
-
Propose two ways of drawing the electron dot formula for Mg.
-
Why does the auditor examine transaction detail for subsidiaries, affiliates, officers, and directors?
-
A powerful new entrant is likely to drive many smaller incumbent firms out of business and their employees out of work. As CEO of a multinational visiting a small country that your firm plans to...
-
A soft drink producer installs a new assembly line to fill 12-oz soda cans. After a week of operation, the plant manager randomly samples 120 cans of soda and weighs the soda. He finds that the soda...
-
Suppose the life of a steel-belted radial tire is uniformly distributed between 30,000 and 45,000 miles. (a) Find the mean tire life. (b) Find the standard deviation of tire life. (c) What percentage...
-
Conduct a five forces analysis of the business school industry or the higher education industry. Identify the strategic group to which your institution belongs. Then use this analysis to explain why...
-
A surge in health insurance premiums imposes an additional burden on a business. A random sample of 10 employees indicates that the average cost increase per employee is about $2,345 with a standard...
-
Use the following data to develop a multiple regression model to predict y from x1 and x2. Discuss the output, including comments about the overall strength of the model, the significance of the...
-
The cost curve for the city water supply is C(Q) = 16 + 1/4 Q2, where Q is the amount of water supplied and C(Q) is the cost of providing Q acre-feet of water. (An acre-foot is the amount of water...
-
A lease agreement between Lennox Leasing Company and Gill Company is described in E21-8. (Round all numbers to the nearest cent.) Refer to the data in E21-8 and do the following for the lessor. (a)...
-
Fieval Leasing Company signs an agreement on January 1, 2010, to lease equipment to Reid Company. The following information relates to this agreement. 1. The term of the non-cancelable lease is 6...
-
Grady Leasing Company signs an agreement on January 1, 2010, to lease equipment to Azure Company. The following information relates to this agreement. 1. The term of the non-cancelable lease is 5...
-
Task Immediate predecessor Task time (min) A B A 12 6 C B 6 B 2 E B 2 F B 12 G C, D 7 H G 5 I E 1 J F, I 4 K H, J 6 L K 7 Demand rate = 140 units/week Assembly operates for 1 shift/day (7 hours), 5...
-
a. What is the difference between moving average and weighted moving average? Using four periods data for January, February, March and April as 100, 104, 95, 99 find the moving average and a weighted...
-
B. A firm has classified its customers in two ways: (1) according to whether or not the account is overdue, and (2) whether the account is new (less than 12 months) or old. An audit of company...

Study smarter with the SolutionInn App


