Based on the general trends in the periodic table, predict which element in each of the following
Question:
Based on the general trends in the periodic table, predict which element in each of the following pairs has the smaller atomic radius:
(a) Na or K
(b) P or N
(c) Ca or Ni
(d) Si or S.
Periodic Table:
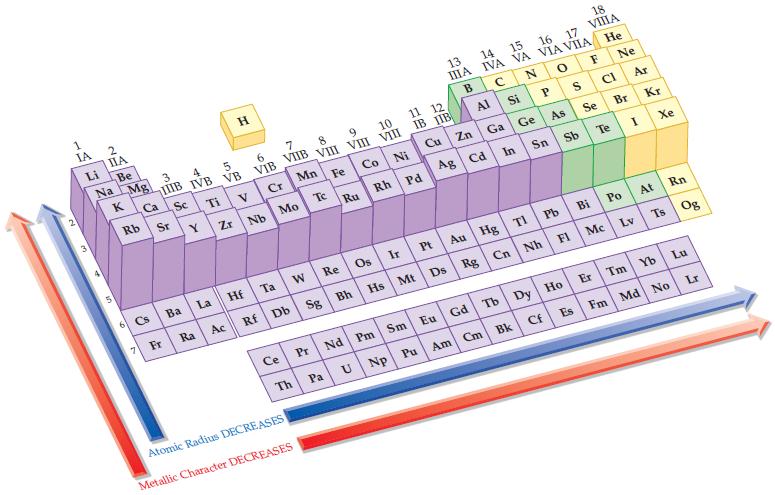
Transcribed Image Text:
1 IA Li 2 IIA Be Na Mg 3 Ca Rb Sr K H 4 5 IIIB IVB VB Sc 6 7 8 9 10 11 12/ VIB VIIB VIII VI VIII IB IIB Tiv Cr Mn Fe Co Ni Zr Nb Mo Tc Cu Ru Rh Pd Y 18 13 14 IIIA 16 17 VIIIA 15 IVA VA VIA VIIA He с NOF Ne B Al Ga Zn Si P S CI Ge Se As Br Ag Cd In Sn Sb Te Ar Atomic Radius DECREASES Metallic Character DECREASES I Kr Xe 6 Cs Ba La Hf Ta W Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn Mc Lv Ts Og 7 Fr Ra Ac Rf Db Sg Bh Hs Mt Ds Rg Cn Nh Fl Am Cm Bk Cf Es Fm Md No Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu Th Pa U Np Pu Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The general trend in atomic radii decreases up a group and across a period from left to righ...View the full answer

Answered By

Jinah Patricia Padilla
Had an experience as an external auditor in Ernst & Young Philippines and currently a Corporate Accountant in a consultancy company providing manpower to a 5-star hotel in Makati, Philippines, Makati Diamond Residences
5.00+
120+ Reviews
150+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Based on the general trends in the periodic table, predict which element in each of the following pairs has the higher ionization energy: (a) Li or Na (b) O or F. Periodic Table: 2 3 4 in 6 3 7 Li...
-
Based on the general trends in the periodic table, predict which element in each of the following pairs has the higher ionization energy: (a) Na or Mg (b) O or S. Periodic Table: 2 3 4 15 6 7 3 11...
-
Based on the general trends in the periodic table, predict which element in each of the following pairs has the most metallic character: (a) Sn or Pb (b) Ag or Sr (c) Al or B (d) Ga or Ge. Periodic...
-
Organizational buyers are ________.
-
Explain how restricted gifts and grants are reported by a public college or university. How would such restricted gifts and grants be recorded and reported by a private college or university?
-
Webster runs a $l00,000 per month cash deficit, requiring periodic transfers from its portfolio of marketable securities. Broker fees are $32 per transaction, and Webster earns 7% on its investment...
-
When we say that a photon is a "quantum of light," what does that mean? What is quantized?
-
Public companies are required to file restated financial statements with the SEC when they discover after the audited financial statements have been issued that the financial statements are...
-
Simplify: 16q42 V Report your answer with a positive value of q.
-
Select the symbol of the element that fits each of the following descriptions: (a) The alkaline earth metal in the sixth period (b) The noble gas in the third period (c) The actinide with the highest...
-
What is the group number for each of the following families of elements? (a) Alkali metals (b) Alkaline earth metals (c) Halogens (d) Noble gases.
-
Suppose you travel \(10 \mathrm{~km}\) from \(\mathrm{P}\) to \(\mathrm{Q}\) at \(10 \mathrm{~km} / \mathrm{h}\) and another \(10 \mathrm{~km}\) from \(\mathrm{Q}\) to \(\mathrm{R}\) at \(20...
-
Explain what is meant by a freemium pricing model. Provide an example of a company, other than one mentioned in this book, that utilizes a freemium model to price its product or service. Do you...
-
What is a board of directors? What is the difference between inside and outside directors?
-
Describe several of the guidelines to setting up a board of advisers.
-
Kim Simpson owns a successful fitness center in an affluent suburb of St. Louis. She just received funding and plans to open six new fitness centers in the St. Louis area over the next two years....
-
Describe the three formal responsibilities of a board of directors.
-
A manager of a car dealership believes there is a relationship between the number of salespeople on duty and the number of cars sold. Suppose the following sample is used to develop a simple...
-
Consider the following cash flows in Table P5.5. (a) Calculate the payback period for each project. (b) Determine whether it is meaningful to calculate a payback period for project D. (c) Assuming...
-
What is the normal procedure for handling the collection of accounts receivable previously written off using the direct write-off method the allowance method?
-
What is imputed interest? In what situations is it necessary to impute an interest rate for notes receivable? What are the considerations in imputing an appropriate interest rate?
-
Indicate three reasons why a company might sell its receivables to another company.
-
Encrypt the message "REDS HACKED BLUE" using the block cipher defined by . (1) = 3 (2)=5 . (3) 1 = (4) = 7 . (5) = 2 (6)=4 (7)=6 Type your answer using UPPERCASE, and omit spaces.
-
solve with explanation please. & (15 points) Apply the critical-path scheduling algorithm with two machines to determ priority hd for the order requirement digraph below and determine the time it...
-
In the beginning, cell phone companies offered data service to their customers' mobile phones and it was billed by the kilobyte. Currently, many customers pay a monthly fee that allows them blocks of...

Study smarter with the SolutionInn App


