Predict which element in each of the following pairs is more electronegative according to the general trends
Question:
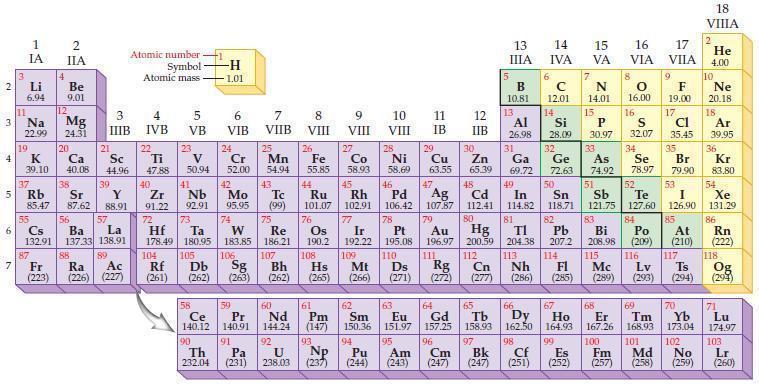
Predict which element in each of the following pairs is more electronegative according to the general trends in the periodic table.
(a) Br or Cl
(b) O or S
(c) Se or As
(d) N or F.
Periodic Table
Transcribed Image Text:
2 3 4 5₁ 6 7 3 11 Li 6.94 1 IA Na 22.99 19 37 al 55 4 87 2 IIA Fr (223) Be 9.01 12 K Ca Sc 39.10 40.08 44.96 Mg 24.31 38 Rb Sr Y 85.47 87.62 88.91 56 57 Cs Ba La 132.91 137.33 138.91 20 21 88 3 IIIB 39 89 Atomic number Symbol - Ac Ra (226) (227) Atomic mass 4 IVB 22 Ti 47.88 40 5 VB 23 V 50.94 41 Zr Nb 91.22 92.91 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 104 105 Rf Db (261) (262) (263) Sg 106 59 Pr 140.91 72 73 W Re Hf Ta 178.49 180.95 183.85 186.21 91 7 VIIB Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 Fe Co 55.85 58.93 Os 190.2 44 45 Ru Rh 101.07 102.91 76 108 9 VIII 61 Pm (147) 27 Bh Hs Mt (262) (265) (266) 93 U NP 238.03 (237) 77 Ir 192.22 109 10 VIII 62 Sm 150.36 94 28 Ni 58.69 46 Pd 106.42 78 11 IB Pt 195.08 110 29 Cu 63.55 13 IIIA 12 IIB 5 B 10.81 13 17 16 VA VIA VIIA 8 6 C 12.01 14 Si 28.09 32 30 31 33 As Se Br Zn Ga Ge 65.39 69.72 72.63 74.92 78.97 79.90 Al 26.98 14 15 IVA 7 N 14.01 15 P 30.97 16.00 83 Bi 208.98 115 16 S 32.07 34 84 Po (209) 47 48 49 50 51 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 79 Au Hg 196.97 200.59 111 112 Rg Cn (271) (272) (277) (286) (285) (289) (293) (294) 81 82 TI Pb 204.38 207.2 113 114 Nh Fl Ds Mc Lv Ts 9 116 F 19.00 Md (258) 17 Cl 35.45 35 53 I 126.90 85 At (210) 66 67 69 70 63 64 65 68 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 95 101 102 Pu Am (244) (243) Cm (247) 97 Bk (247) 98 99 100 Cf Es Fm (251) (252) (257) 117 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a Br or Cl Cl is more electronegative Electronegativity increases increasing from left to righ...View the full answer

Answered By

Zablon Gicharu
I am an educator who possesses the requisite skills and knowledge due to interacting with students for an extended period. I provide solutions to various problems in step-by-step explanations, a well-thought approach and an understandable breakdown. My goal is to impart more straightforward methodologies and understanding to students for more remarkable achievements.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Predict which element in each of the following pairs is more electronegative according to the general trends in the periodic table. (a) Se or Br (b) C or B (c) Te or S (d) Ba or Be. Periodic Table 2...
-
Predict which element in each of the following pairs is more electronegative according to the general trends in the periodic table: (a) H or Cl (b) Br or I (c) P or S (d) As or Sb.
-
Predict which element in each of the following pairs is more electronegative according to the general electronegativity trends in the periodic table: (a) N or O (b) Br or Se (c) F or Cl (d) Si or C.
-
ABC Client Preliminary Analytics - PBC Trial Balance December 31, 2019 Scope: Performance Materiality $210,000 and 10% 12/31/19 Balance 12/31/18 Account Class Balance Variance * Varlance Cash...
-
List the required governmental fund and proprietary fund financial statements under GASB 34. On what basis of accounting are these statements prepared?
-
Express the confidence interval (0.0169, 0.143) in the form of p E < p < p + E. In express the confidence interval using the indicated format. (The confidence intervals are based on the proportions...
-
In Figure P27.28, an external magnetic field is directed upward throughout a region that contains four currentcarrying wires having the lengths and currents shown. Rank the wires according to the...
-
1. Do Sanders, Ono, and Lyons have the right to challenge the annual picnics value in communicating the corporate culture values? 2. How would company management at Exterior Lighting know whether the...
-
18. The atomic number of elements P, Q, R and T are 19,17, 14 and 6 respectively. The pair of elements that can react to form an ionic compound is A. Q and T B. R and Q C. Q and P D. R and T 19. The...
-
Which elements are more electronegative: semimetals or nonmetals?
-
Write the electron dot formula and draw the structural formula for each of the following polyatomic ions. (a) PH 4 + (b) SeO 3 2 (c) CO 3 2 (d) BO 3 3
-
The following information is known about a project a. Draw the network diagram for this project. b. Determine the critical path and project duration c. Calculate the slack for eachactivity Activity...
-
All financial institutions, to varying degrees, are exposed to credit, interest rate, liquidity, foreign exchange, and operating risks. Briefly and clearly define each.
-
For a three year bond of $2,400 at a simple interest rate of 10% per year, find the semiannual interest payment and the total interest earned over the life of the bond. 1.) The semiannual interest on...
-
How is the COGS Inventory calculated? Invoice No. Date 12/03/18 Sales Journal - Credit Sales Accounts Debited 1201 Beverly's Building Products 12/03/18 1202 Bilder Construction Co. 12/04/18 1203...
-
The price of capital (r) is $20. 180 in of cap 160 140 [120 100- 80 60 40 20 0 20 40 80 100 120 140 160 180 Units of labor d) At point A, the Marginal Product of Labor (MPL) is 240 and the Marginal...
-
Will the expected value of a risky asset be higher or lower under a risk-neutral distribution than under the physical distribution? Why?
-
Honre Corporation's warehouse and Filip Company's office building were located side by side until a fire raced through both structures, completely destroying them. The warehouse has an adjusted basis...
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
NPV and Collection time your firm has an average receipt size of $95. A ban has approached you concerning a lockbox service that will decrease your total collection time by two days. You typically...
-
Using weighted average Delay a mail-order firm processes 4,500 checks per month. Of these, 60 percent are for $50 and 40 percent are for $70. The $50 checks are delayed two days on average; the $70...
-
Value of Lockboxes paper submarine Manufacturing is investigating a lockbox system to reduce its collection time. It has determined the following. The total collection time will be reduced by three...
-
Fanny Farmer held an annual auction at her farm to sell tools, animals, and leftover crops. At the auction there was no indication given whether the auction was with or without reserve. Bob bid $ 5 0...
-
What resistance to change do the employees in the supply company appear to be exhibiting? Support your answer. Forces resisting organizational change include the following: Direct Costs, Saving Face,...
-
'STAR' Technique Situation Describe the situation that you were involved in . Provide a detailed background and context, answer "where" and" when". Task What were you trying to achieve in the...

Study smarter with the SolutionInn App


