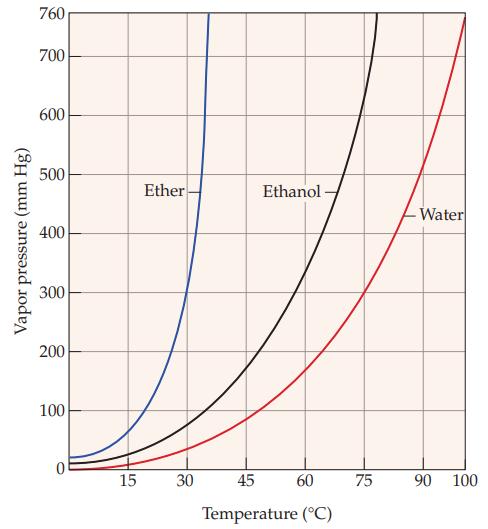
Refer to Figure 11.5 and determine the approximate boiling point of ethanol. Figure 11.5 Vapor pressure (mm
Question:
Refer to Figure 11.5 and determine the approximate boiling point of ethanol.
Figure 11.5
Transcribed Image Text:
Vapor pressure (mm Hg) 760 700 600 500 400 300 200 100 15 Ether 30 Ethanol 45 60 Temperature (°C) 75 Water 90 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
80 C The ...View the full answer

Answered By

Ashok Kumar Malhotra
Chartered Accountant - Accounting and Management Accounting for 15 years.
QuickBooks Online - Certified ProAdvisor (Advance - QuickBooks Online for 3 years.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to Figure 11.5 and determine the approximate boiling point of ether. Figure 11.5 Vapor pressure (mm Hg) 760 700 600 500 400 300 200 100 0 15 Ether 30 Ethanol 45 60 Temperature (C) 75 > -Water...
-
What is oral Language? Why is oral Language important? What are three ways educators can Support oral Language? What are three ways Parents can support oral Language at home? What are three resources...
-
Refer to Figure 11.5 and determine the boiling point of ethanol at an elevation where the atmospheric pressure is 0.500 atm. Figure 11.5 Vapor pressure (mm Hg) 760 700 600 500 400 300 200 100 0 15...
-
Following are financial statements (historical and forecasted) for the Global Products Corporation. A. Assume that the cash account includes only required cash. Determine the dollar amount of equity...
-
Are lessees more likely to be in higher or lower income tax brackets than lessors?
-
In the chapter, you saw how to create a production possibilities frontier for the United States and Mexico. Lets take a look at how to combine these PPFs to make one PPF for the U.S.Mexico trade...
-
Express the magnetic field due to a uniformly moving charged particle in terms of the electric field of the particle and its velocity.
-
Indicate whether each of the following transactions would increase (+), decrease(??), or not affect (N/A) total assets, total liabilities, and total stockholders??equity. Stockholders' Transaction...
-
Kazibwe, a businessman does not maintain a complete set of books for his business transactions. The following is a summary of his cash transactions for the year ended December 31, 2015. Receipts Kshs...
-
Which of the following are observed general properties of liquids? (a) Fixed volume (b) Mix homogeneously (c) Compress significantly (d) More dense than gases.
-
Which of the following is an observed property of liquids? (a) Liquids are more dense than gases. (b) Liquids expand and compress significantly. (c) Liquids have a fixed shape and variable volume....
-
Suppose you bought a bond with an annual coupon rate of 6.5 percent one year ago for $1,032. The bond sells for $1,020 today. a. Assuming a $1,000 face value, what was your total dollar return on...
-
Write an x86 assembly program that computes the determinant of a matrix composed of signed 16-bit integers, with the following requirements: 1. The size of the matrix is 2x2. 2. The matrix will be...
-
Solve the steady-state temperature distribution which satisfies Laplace's equation +=0 by Liebmann's iteration method (until fourth iteration) satisfying the following boundary conditions: u(0, y) =0...
-
Suppose the pre-acquisition market capitalization of Amersham was $8 billion and that this market capitalization did not reflect the possibility that Amersham will be acquired. In order to justify...
-
-Convertible Preferred Stock -TG pre money: 5M -TG post money: 9M Red Baron: -Convertible Participating -Preferred Stock -RB pre money: 6M -RB post money: 12M -capped at 2.5 Question: HOW TO DRAW THE...
-
18. Find grammars for the following languages on = {a}. a. L = {w: |w| mod 3 >0}. b. L= {w: w mod 3 = 2}.
-
Tommi inherits Dierhopf Corporation common stock from her uncle, Norvel. Norvels adjusted basis in the stock is $200,000, and the fair market value is $380,000. Six months after Norvels death, the...
-
Suppose that a flow network G = (V, E) violates the assumption that the network contains a path s t for all vertices V. Let u be a vertex for which there is no path s u t. Show that there must...
-
Digital Organics (DO) has the opportunity to invest $1 million now ( ) and expects after-tax returns of $600,000 in and $700,000 in . The project will last for two years only. The appropriate cost of...
-
You are considering a five-year lease of office space for R&D personnel. Once signed, the lease cannot be canceled. It would commit your firm to six annual $100,000 payments, with the first payment...
-
Consider another perpetual project like the crusher described in Section 19.1. Its initial investment is $1,000,000, and the expected cash inflow is $85,000 a year in perpetuity. The opportunity cost...
-
CathFoods will release a new range of candies which contain anti-oxidants. New equipment to manufacture the candy will cost $3 million, which will be depreciated by straight-line depreciation over...
-
Kebt Corporation's Class Semi bonds have a 14-year maturity and a 10.25% coupon paid semiannually (5.125% each 6 months), and those bonds sell at their $1,000 par value. The firm's Class Ann bonds...
-
Sheet Company reported the following net income and dividends for the years indicated: Year 20X5 20X6 20X7 Net Income $ 35,000 45,000 30,000 Dividends $ 12,000 20,000 14,000 Pillow Corporation...

Study smarter with the SolutionInn App


