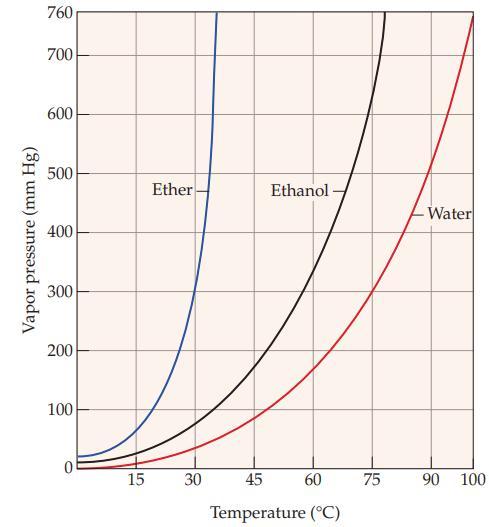
Refer to Figure 11.5 and determine the boiling point of ethanol at an elevation where the atmospheric
Question:
Refer to Figure 11.5 and determine the boiling point of ethanol at an elevation where the atmospheric pressure is 0.500 atm.
Figure 11.5
Transcribed Image Text:
Vapor pressure (mm Hg) 760 700 600 500 400 300 200 100 0 15 Ether 30 Ethanol- 45 60 Temperature (°C) 75 Water 90 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)
The boiling poin...View the full answer

Answered By

ZIPPORAH KISIO LUNGI
I have worked on several other sites for more than five years, and I always handle clients work with due diligence and professionalism. Am versed with adequate experience in the fields mentioned above in which have delivered quality papers in research, thesis, essays, blog articles, and so forth.
I have gained extensive experience in assisting students to acquire top grades in biological, business and IT papers. Notwithstanding that, I have 7+ years of experience in corporate world software design and development.
5.00+
194+ Reviews
341+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to Figure 11.5 and determine the boiling point of water at an elevation where the atmospheric pressure is 0.500 atm. Figure 11.5 Vapor pressure (mm Hg) 760 700 600 500 400 300 200 100 0 15...
-
Using the vapor-pressure curves in Figure 11.25, (a) Estimate the boiling point of ethanol at an external pressure of 200 torr; (b) Estimate the external pressure at which ethanol will boil at 60 oC;...
-
The normal boiling point for acetone is 56.5oC. At an elevation of 5300 ft, the atmospheric pressure is 630 torr. What would be the boiling point of acetone (Hvap = 32.0 kJ/mol) at this elevation?...
-
On January 1, 2011, M Company granted 98,000 stock options to certain executives. The options are exercisable no sooner than December 31, 2013 and expire on January 1, 2017. Each option can be...
-
A summary of changes in Pen Corporation's Investment in Sam account from January 1, 2011, to December 31, 2013, follows (in thousands): ADDITIONAL INFORMATION1. Pen acquired its 80 percent interest...
-
The uniform crate resting on the dolly has a mass of 500 kg and mass center at G. If the front casters contact a high step, and the coefficient of static friction between the crate and the dolly is ...
-
A conducting bar of width \(w=0.12 \mathrm{~m}\) and mass \(m=\) \(8.0 \mathrm{~g}\) can slide freely on two parallel conducting rails positioned at an incline of \(\theta=15^{\circ}\) (Figure...
-
D-List Calendar Co. specializes in manufacturing calendars that depict obscure comedians. The company uses a standard cost system to control its costs. During one month of operations, the direct...
-
Question 2) Suppose there were an alternative replacement machine that could do the same job as the new stamping machine under your consideration in Question 1. The alternate choice offers the...
-
Draw the electron dot formula for H 2 S. How many nonbonding electron pairs are in a hydrogen sulfide molecule? H S
-
Calculate the percentage of water in each of the following hydrates. (a) SrCl 2 6 H 2 O (b) K 2 Cr 2 O 7 2 H 2 O (c) Co(CN) 3 3 H 2 O (d) Na 2 CrO 4 4 H 2 O.
-
Fin u v. (a) u = (2, 3), v = (5, - 7) (b) u = (1, -5,)4, v = (3, 3, 3)
-
A surveyed plat of land a shown. Elevations are recorded on the stakes at the grid corners. The grid squares are 100 ft. 1. Draw the contour lines at 5' intervals. (HW #1) 2. Assuming the entire area...
-
To reduce the total mass of the assembly shown in the figure, a new design is being considered in which the diameter of shaft BC will be smaller. Determine the smallest diameter of shaft BC for which...
-
The Centers for Medicare & Medicaid Services (CMS) and other payers plan to shift to upside risk. What is the definition of upside risk?
-
11. Generate the requested statistics based on the figures provided in the following table. Type your answers to the calculations in Table 9 of your data file Remember to show your calculations....
-
10 You can show that A = and B = 0 -1 diagonalization. First, find their common diagonalization D. D = are similar matrices using the fact that they have the same Now find a matrix Pl satisfying...
-
In determining the amount of a realized gain or loss to be recognized in the current year, certain types of gains and losses are deferred, whereas others are disallowed. What is the difference...
-
The relationship described in question 7 does not always appear to hold. What factors, besides the number of firms in the market, might affect margins?
-
Suppose that after-tax investment is defined as the investment outlay minus the present value of future depreciation tax shields. In the guano project, for example, after-tax investment would be...
-
Here are inflation rates and stock market and Treasury bill returns between 1996 and 2000: a. What was the real return on the S&P 500 in each year? b. What was the average real return? c. What was...
-
An oil company executive is considering investing $10 million in one or both of two wells: Well 1 is expected to produce oil worth $3 million a year for 10 years; well 2 is expected to produce $2...
-
Briefly explain the reasons for the formation of monopolies in digital products.
-
The price elasticity of toy cars that you sell is 4.00; you currently charge a price of $9.00 and the marginal cost of toy cars is $8.00. a. Calculate the marginal revenue from the given information...
-
membership with Junktrader, which provides them advance notice of potentially attractive offers. a. Junktrader provided online advertising services for another company for $380 on account. b. On the...

Study smarter with the SolutionInn App


