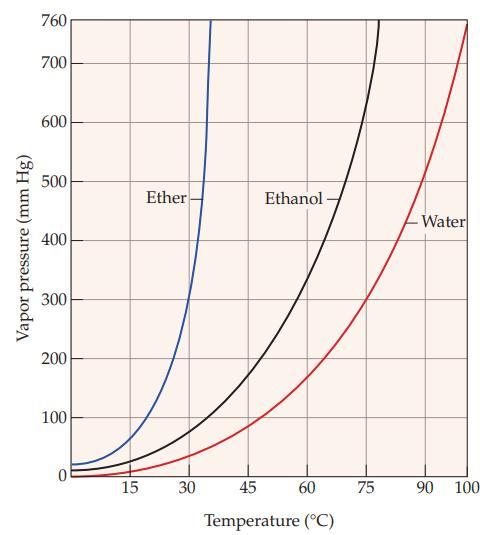
Refer to Figure 11.5 and estimate the vapor pressure of ether at each of the following temperatures:
Question:
Refer to Figure 11.5 and estimate the vapor pressure of ether at each of the following temperatures:
(a) 15 °C
(b) 30 °C
Figure 11.5
Transcribed Image Text:
Vapor pressure (mm Hg) 760 700 600 500 400 300 200 100 0 15 Ether 30 Ethanol- 45 60 Temperature (°C) 75 -Water 90 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The vapor pressure of ether at 15 C is approximately 300 mmHg ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to Figure 11.5 and estimate the vapor pressure of ethanol at each of the following temperatures: (a) 45 C (b) 60 C Figure 11.5 Vapor pressure (mm Hg) 760 700 600 500 400 300 200 100 0 15 Ether...
-
The vapor pressure of water at various temperatures follows: (a) Draw a scatter diagram of these data. What type of relationship seems appropriate in relating y to x? (b) Fit a simple linear...
-
The vapor pressure of 1-chlorotetradecane at several temperatures is tabulated here. (a) Use two-point linear interpolation to estimate the value of p* at T = 185 o C. (b) Write a computer subroutine...
-
Explain what you would assume the yield curve would look like during economic expansion and why.
-
Cite the conditions under which you would expect the balance of an equity investment account on a balance sheet date subsequent to acquisition to be equal to the underlying book value represented by...
-
Two discus throwers keep a record of their best throws (in metres) in the last ten competitions. As a coach, you can only choose one of them for the next competition. Which would you choose? Justify...
-
The space to the right of the \(y\) axis in Figure P29.28 contains a uniform magnetic field of unknown magnitude that points in the positive \(z\) direction. As a conducting square loop placed in the...
-
Roman Products, Inc., is a wholesaler of mens hair products. The company began operations on January 1, 2010. The following transactions relate to securities acquired by Roman Products, Inc., which...
-
a) Money markets are used to trade debt securities and instruments with maturities of less than one year. Identify three characteristics of the money market. (6 marks) b) Explain four reasons for the...
-
What is the general relationship between the boiling point of a liquid and its vapor pressure?
-
What is the general relationship between the vapor pressure of a liquid and its temperature?
-
Can it be ethically justified for a manufacturer to sell a product with a warranty less than the law requires?
-
Solve the initial value problem. dt =-64 sin 8t- 1000 OA. s= sin 8t- OB. s= sin 8t- (1 3x 2 OD. s= sin 8t- 2 2 OC. s 8 cos 8t- 2 2 s'(0)=6, s(0) = -2 +5t-2 THE -3 +6 +61-3
-
How much will I need to deposit into my account at the beginning of each month for the next 50 years if I want $2,700,000 at the end, assuming my account makes 6% pa compounded quarterly over that...
-
JanehasaskedyoutomakechangestotheCompanySettingstoset upanduseadditionalQuickBooksfeatures. Jane knows that discountsare a part of the business. She asks you to make sure that when you add...
-
A couple who just had a girl decide to save up to pay the fees. of the child's university. It can create a university fund which bears interest on a annual rate of 7% assuming the child is starting...
-
Variable costs related to sales equal 70% of the selling price and the company has fixed expenses estimated at R400 000 every month. The company had a cash balance of R200 000 at the end of May 2020....
-
Assume that in problem 37, the United Express Company sells a truck that cost $60,000 in 2011 for $15,000 in June 2014. Assume that none of the truck was expensed in 2011. Compute the adjusted basis...
-
How do network effects help Facebook fend off smaller social-networking rivals? Could an online retailer doing half as much business compete on an equal footing with Amazon in terms of costs? Explain.
-
Which of the following features would increase the value of a corporate bond? Which would reduce its value? a. The borrower has the option to repay the loan before maturity. b. The bond is...
-
The shareholders of the Pickwick Paper Company need to elect five directors. There are 200,000 shares outstanding. How many shares do you need to own to ensure that you can elect at least one...
-
Can you think of any new kinds of security that might appeal to investors? Why do you think they have not been issued?
-
Write a C program to capitalize the first character in each sentence from a given file. Assume a sentence ends with a period '.' To get the result, the execution of this program must be cooperated...
-
Assessment 2 Leaders are Born, not made. (25% of Total Semester marks) Description and Purpose Topics to be covered: Purpose: The purpose of this assignment is to allow students to critically...
-
An all-equity firm, Poly Inc., is considering the following projects: Proposed Project Beta IRR A 0.2 7.5% B 0.5 8.0% C 0.8 11.8% D 1.0 12.3% E 1.1 12.5% F 1.5 18.0% The beta of the firm's stock is...

Study smarter with the SolutionInn App


