Refer to the periodic table and state the highest energy sublevel in a silver atom. Periodic Table:
Question:
Refer to the periodic table and state the highest energy sublevel in a silver atom.
Periodic Table:
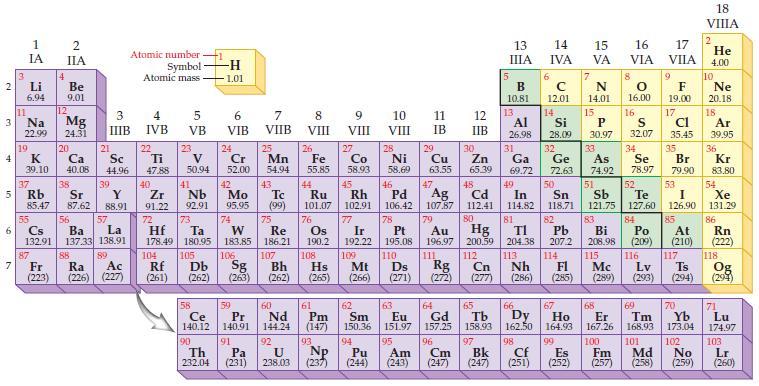
Transcribed Image Text:
2 3 4 10 6 3 7 11 1 IA Li 6.94 Na 22.99 19 37 5 Rb K 39.10 4 87 2 IIA Fr (223) Be 9.01 12 Mg 24.31 20 38 Ca Sc 40.08 44.96 21 Sr Y 85.47 87.62 88.91 55 56 La Cs Ba 132.91 137.33 138.91 88 3 4 IIIB IVB 39 57 89 Atomic number Symbol Atomic mass Ac Ra (226) (227) 22 Ti 47.88 5 VB 23 104 V 50.94 40 Nb Zr 91.22 92.91 41 105 -H 1.01 6 VIB 58 Ce 140.12 90 Th 232.04 24 Cr 52.00 42 Mo 95.95 74 72 73 W Re Hf Ta 178.49 180.95 183.85 186.21 106 Rf Db Sg (261) (262) (263) 59 Pr 140.91 91 7 VIIB Pa (231) 25 Mn 54.94 43 Tc (99) 75 107 60 Nd 144.24 8 VIII 92 26 27 Fe Co 55.85 58.93 Bh Hs (262) (265) 44 45 Ru Rh 101.07 102.91 76 Os 190.2 108 61 Pm (147) 9 VIII 93 U NP 238.03 (237) 77 Ir 192.22 109 Mt (266) 62 Sm 150.36 10 VIII 28 Ni 58.69 46 Pd 106.42 78 11 IB 94 95 Pu Am (244) (243) 13 IIIA 12 IIB 5 6 7 C 12.01 14 N 14.01 15 29 30 31 34 Si P 28.09 30.97 32 33 Cu Zn Ga Ge As Se 63.55 65.39 69.72 72.63 74.92 78.97 48 49 50 52 Ag Cd In Sn Sb Te 107.87 112.41 114.82 118.71 121.75 127.60 80 47 51 B 10.81 13 Al 26.98 14 15 IVA 16 VA VIA 79 81 82 Pt Au Hg TI Pb 195.08 196.97 200.59 204.38 207.2 110 111 112 113 114 Ds Rg Cn Nh Fl (271) (272) (277) (286) 83 Bi 208.98 115 16.00 16 52 S 32.07 84 Po (209) 17 VIIA 9 Md (258) F 19.00 17 Cl 35.45 35 Br 79.90 53 I 126.90 85 At (210) 116 Mc Lv (285) (289) (293) (294) Ts 117 66 70 63 64 65 67 68 69 Eu Gd Tb Dy Но Er Tm Yb 151.97 157.25 158.93 162.50 164.93 167.26 168.93 173.04 96 98 99 100 Cf Es Fm (251) (252) (257) 101 102 Cm (247) 97 Bk (247) 18. VIIIA 2 He 4.00 10 Ne 20.18 18 Ar 39.95 36 Kr 83.80 54 Xe 131.29 86 Rn (222) 118 Og (294) 71 Lu 174.97 103 No Lr (259) (260)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
Because Ag is ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the periodic table and state the highest energy sublevel for each of the following elements. (a) He (b) K (c) U (d) Pd (e) Be (f) Co (g) Si (h) Pt. Periodic Table: 2 3 4 10 6 3 7 11 Li 6.94...
-
Refer to the periodic table and state the highest energy sublevel for each of the following elements. (a) H (b) Na (c) Sm (d) Br (e) Sr (f) C (g) Sn (h) Cs. Periodic Table: 2 3 4 10 6 3 7 11 Li 6.94...
-
Element 43 is used in medical radiology to locate tumors. Refer to the periodic table and state whether Tc has any stable isotopes. Periodic Table: 2 3 4 5 6 7 3 11 1 IA Li Na 19 37 Rb 55 87 Fr 4 2...
-
Show that if powers of x greater than x 5 are neglected. In sin x X =-=-x-x 180
-
Private College Transactions. Elizabeth College, a small private college, had the following transactions in fiscal year 2011. 1. Billings for tuition and fees totaled $5,600,000. Tuition waivers and...
-
Perform a common size analysis and percentage change analysis. What do these analyses tell you about Computron? The first part of the case, presented in Chapter 7, discussed the situation that...
-
It The wavelengths of light emitted by a firefly span the visible spectrum but have maximum intensity near \(550 \mathrm{~nm}\). A typical flash lasts for \(100 \mathrm{~ms}\) and has a power of...
-
ProCom Industries operates in several different industries. Total sales for ProCom are $14,000,000, and total common costs are $6,500,000 for 2011. For internal reporting purposes, ProCom allocates...
-
A booking agent estimates the 1/7 of the gigs she books are for private parties and 4/15 of the gigs she books are for public performance where her bands are the opening acts. What fraction of the...
-
Before Moseleys discovery in 1913, the periodic law stated that physical and chemical properties tend to repeat periodically when elements are arranged according to what trend?
-
Predict which of the following metals has chemical properties most similar to zinc: Fe, Cu, or Cd.
-
Repeat for the 20082009 NFL seasons. In exercise, the home and away attendance figures for the National Football League for the 20122013 seasons were recorded. a. Analyze the relationship between the...
-
Describe what is meant by the term core strategy and why it is important.
-
What is a geographic expansion strategy, and what are the keys to implementing a successful geographic expansion strategy for an entrepreneurial firm?
-
Compute the average cost of 1 repair. Construct an 80 % confidence interval. An automobile manufacturer wants to study the repair record of its own cars. The performance of 1,000 cars and their...
-
What are several rules of thumb to follow for selling products overseas?
-
Describe the characteristics of a fragmented industry. What is the primary opportunity for new firms in fragmented industries?
-
Use a computer to develop the equation of the regression model for the following data. Comment on the regression coefficients. Determine the predicted value of y for x1 = 200 and x =7. ! 3 9 4 8 9...
-
The column shown in the figure is fixed at the base and free at the upper end. A compressive load P acts at the top of the column with an eccentricity e from the axis of the column. Beginning with...
-
Springsteen Co. had the following activity in its most recent year of operations. (a) Pension expense exceeds amount funded. (b) Redemption of bonds payable. (c) Sale of building at book value. (d)...
-
Each of the following items must be considered in preparing a statement of cash flows (indirect method) for Grander son Inc. for the year ended December 31, 2010. (a) Plant assets that had cost...
-
The income statement of Rodriquez Company is shown below. Additional information: 1. Accounts receivable decreased $310,000 during the year. 2. Prepaid expenses increased $170,000 during the year. 3....
-
Do you know your credit score? Have you ever checked your credit report? Do you know who maintains your credit information? Is your credit score important to you over the next 3-5 years, do you...
-
A firm has a payable of C$ 4,000,000.00. They hedge this exposure with a forward participation contract with a guaranteed rate of $1.5500/C$ and a participation rate of 25%. If at the time of payment...
-
What are bond ratings and why are they assigned? Bonds are like loans the issuers get from investors? Just as your credit worthiness is evaluated when you apply for a loan, the credit worthiness of...

Study smarter with the SolutionInn App


