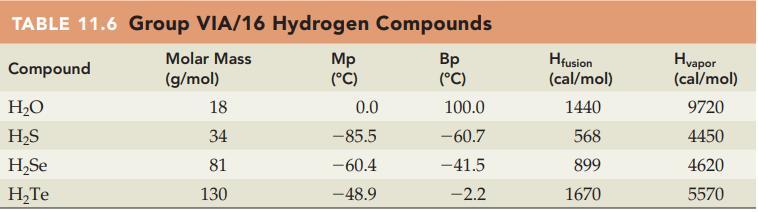
Refer to the trends in Table 11.6 and estimate a value for the physical properties of radioactive
Question:
Refer to the trends in Table 11.6 and estimate a value for the physical properties of radioactive H2Po, that is, predict a value for the Mp, Bp, Hfusion, and Hvapor.
Table 11.6
Transcribed Image Text:
TABLE 11.6 Group VIA/16 Hydrogen Compounds Molar Mass Compound (g/mol) H₂O H₂S H₂Se H₂Te 18 34 81 130 Mp (°C) 0.0 -85.5 -60.4 -48.9 Bp (°C) 100.0 -60.7 -41.5 -2.2 Hfusion (cal/mol) 1440 568 899 1670 Hvapor (cal/mol) 9720 4450 4620 5570
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
From the table we can observe a trend in the properties of Group VIA16 Hydrogen Compounds As we move ...View the full answer

Answered By

Payal Mittal
I specialize in finance and accounts.You can ask any question related to til undergradution.Organizational behaviour and HRM are my favourites for you can always relate to them and is an art with practical knowledge base.
4.90+
226+ Reviews
778+ Question Solved
Related Book For 

Introductory Chemistry Concepts And Critical Thinking
ISBN: 9780321804907
7th Edition
Authors: Charles Corwin
Question Posted:
Students also viewed these Sciences questions
-
Refer to the Trends in Biomaterials & Artificial Organs (Jan. 2003) study of a new bonding adhesive for teeth, presented in Exercise. Recall that the new adhesive (called Smartbond) has been...
-
Without referring to Table 11.6, predict which compound in each of the following pairs has the higher heat of vaporization (cal/mol): (a) H 2 O or H 2 Se (b) H 2 S or H 2 Te. Table 11.6 TABLE 11.6...
-
Without referring to Table 11.6, predict which compound in each of the following pairs has the higher heat of fusion (cal/mol): (a) H 2 O or H 2 S (b) H 2 S or H 2 Se. Table 11.6 TABLE 11.6 Group...
-
The e-commerce business in China has entered a golden period, with transaction volume of online trading reaching 21.86 billion yuan (US$2.64 billion) in 2004. With 94 million Internet users, more...
-
Pal Corporation purchased for cash 6,000 shares of voting common stock of Sap Corporation at $16 per share on July 1, 2011. On this date, Saps equity consisted of $100,000 of $10 par capital stock,...
-
Determine the force P that must be applied perpendicular to the handle in order to hold the mechanism in equilibrium for any angle of rod CD. There is a couple moment M applied to the link BA. A...
-
The magnitude of the magnetic field in a magnetic resonance imaging (MRI) machine can be as great as \(B=3.0 \mathrm{~T}\). Under normal circumstances, this field cannot be shut off by just flipping...
-
The Sports Equipment Division of Brandon McCarthy Company is operated as a profit center. Sales for the division were budgeted for 2010 at $900,000. The only variable costs budgeted for the division...
-
"Managing Away Bad Habits Team Assignment Organizational Behavior IILeadership Assigned is ashort case from the exercise Managing Away Bad Habits. The task is to develop a turnaround strategy for...
-
Write a balanced equation for the reaction of hydrogen and oxygen gases.
-
Complete and balance the following equations. (a) Li(s) + H 2 O(l) S (b) Na 2 O(s) + H 2 O(l) S (c) CO 2 (g) + H 2 O(l) S
-
How might the creditworthiness of a new customer be checked?
-
In scenario A, the formula to calculate the future value after 20 years is indeed Ct = PV (1 + r)20. This formula considers 20 compounding periods because the interest is applied annually. Each year,...
-
A QAM signal has the mathematical form given below: Ym (t) = Im.cos(wet) - Qm. sin(wet) ym (t) = Am1.cos(wet + m2) Where: Im and Qm are the in-phase carrier weightings (cos() carrier) and quadrature...
-
Derive the Time Schrodinger wave equation in reference to the general partial differential wave equation.
-
(a) Simplify the following summation to produce a closed form. Show your work, clearly stating when you apply each summation identity. ) (b) Convert the following recurrence into a summation by...
-
what is the write code to add event handler to button in this code ? Click Me! var button = document.getElementsByTagName("button"); button.onclick= function () { alert("Click fired!"); } var button...
-
During 2011, Schottenheim Corporation buys 20 laptop computers and a mainframe computer to use in its general sales offices. Schottenheim buys 14 laptops for $42,000 on March 29, 6 laptops for...
-
Gopher, Inc. developing its upcoming budgeted Costs of Quality (COQ) with the following information: Expense Item Budget Raw Materials Inspection $ 15,000 EPA Fine 200,000 Design Engineering 15,000...
-
Amalgamated Products has three operating divisions: To estimate the cost of capital for each division, Amalgamated has identified the following three principal competitors: Assume these betas are...
-
Look at Table 9.2. What would the four countries? betas be if the correlation coefficient for each was 0.5? Do the calculation and explain. Ratio of Standard Correlation Betat Coefficient Deviations*...
-
Investors home country bias is diminishing rapidly. Sooner or later most investors will hold the world market portfolio, or a close approximation to it. Suppose that statement is correct. What are...
-
The city of New Coventry decides to elect a mayor using the Electoral College weighted voting system, with the voters being the three boroughs of the city. Each borough is awarded two city Senators...
-
Tunnel inspections can be expensive, operationally difficult, with a possible loss of revenue during the inspection. The result of these approaches is that tunnel inspections get delayed, postponed...
-
Howard Company is considering three financing plans: all equity; 60 per cent equity and 40 per centdebt; and 40 per cent equity and 60 per cent debt. Total funds needed are Rs 300,000. EBIT is...

Study smarter with the SolutionInn App


