If y = u(x)v(x), prove that Hence prove Leibnizs theorem for the nth derivative of a product:
Question:
If y = u(x)v(x), prove that
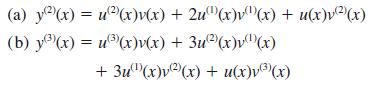
Hence prove Leibniz’s theorem for the nth derivative of a product:
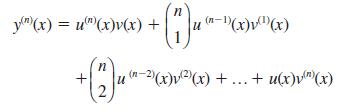
Transcribed Image Text:
(a) y(x) = u(x)v(x) + 2u(x)v(x) + u(x)v)(x) (b) y(x) = u(x)v(x) + 3u2(x)v(x) + 3u(x)v(x) + u(x)v(x)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a b y uxvx...View the full answer

Answered By

Keziah Thiga
I am a self motivated financial professional knowledgeable in; preparation of financial reports, reconciling and managing accounts, maintaining cash flows, budgets, among other financial reports. I possess strong analytical skills with high attention to detail and accuracy. I am able to act quickly and effectively when dealing with challenging situations. I have the ability to form positive relationships with colleagues and I believe that team work is great key to performance. I always deliver quality, detailed, original (0% plagirism), well-researched and critically analyzed papers.
4.90+
1504+ Reviews
2898+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use Leibnizs theorem (Question 71) to find the following: Data from Question 71 If y = u(x)v(x), prove that Hence prove Leibnizs theorem for the nth derivative of a product: (a) d dxs (x sin x) (put...
-
One of several Leibniz Rules in calculus deals with higher-order derivatives of products. Let (fg) (n) denote the nth derivative of the product fg, for n 1. a. Prove that (fg) (2) = f"g + 2 f'g' +...
-
Suppose the European Parliament is considering legislation that will decrease tariffs on agricultural products imported from the United States. The benefit of the tariff reduction is estimated at 5...
-
Find the directive gain and directivity of the small loop antenna.
-
A steel tank contains 300 g of ammonia gas (NH3) at a pressure of 1.35 x 106 Pa and a temperature of 77oC. (a) What is the volume of the tank in liters? (b) Later the temperature is 22oC and the...
-
Two metal balls have charges +q and q. How will the force on one of them change if? (a) The balls are placed in water, the distance between them being unchanged, and (b) A third uncharged metal ball...
-
What mix of inputs should be used to produce a given level of output?
-
Rickys Repair Shop has a monthly target profit of $17,000. Variable costs are 60% of sales, and monthly fixed costs are $8,000. Requirements 1. Compute the monthly margin of safety in dollars if the...
-
Keller Company makes two models of battery-operated boats, the Sandy Beach and the Rocky River. Basic production information follows: Sandy Beach Direct materials cost per unit Direct labor cost per...
-
Find the radius of curvature at the point (2, 8) on the curve y = x 3 .
-
Prove that where d" - (e sin bx) = (a + b)/ex sin(bx + n0) d.x"
-
Sketch the following regions and write an iterated integral of a continuous function f over the region. Use the order dy dx. R = {(x, y): 1 x 2 x + 1 y 2x + 4}
-
Provide realistic examples of how a requirements document may be: a. ambiguous b. incorrect c. incomplete d. unverifiable
-
A young girl and her mother are swinging on a swing set. Who, if either, has the longer period of oscillation? Explain.
-
Scuba divers are warned that if they must make a rapid ascent, they should exhale on the way up. If a diver rapidly ascends to the surface with lungs full of air, his lungs could be damaged. Explain...
-
Classify these Bluetooth layers using the OSI model: a. baseband; b. L2CAP; c. RFCOMM.
-
Briefly describe the differences between the waterfall and spiral development models.
-
Hogyoku Tamukeyama owns and operates a successful dry cleaning business in Bangor, Maine. Summary financial data for a typical month of operations are as follows: Because it is the end of winter,...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
Consider the following mechanism, which results in the formation of product P: If only the species A is present at t = 0, what is the expression for the concentration of P as a function of time? You...
-
Using the preequilibrium approximation, derive the predicted rate law expression for the following mechanism: A+B- P
-
For the reaction I (aq) + OCl (aq) OI (aq) + Cl (aq) occurring in aqueous solution, the following mechanism has been proposed: a. Derive the rate law expression for this reaction based on this...
-
Delph Company uses job-order costing with a plantwide predetermined overhead rate based on machine-hours. At the beginning of the year, the company estimated that 55,000 machine-hours would be...
-
The Assembly Department of Interface, Inc., manufacturer of computers, had 500 units of beginning inventory in September, and 4,000 units were transferred to it from the Production Department. The...
-
Shea Furniture started and finished Job 310 during December. The company's records show that the following direct materials were requisitioned for Job 310: (Click the icon to view the direct...

Study smarter with the SolutionInn App


