Refer to the energy level diagram for Ar+ given in Figure 7.6 and find the spinorbit coupling
Question:
Refer to the energy level diagram for Ar+ given in Figure 7.6 and find the spin–orbit coupling constant A for the ground state 2P and excited state 4D terms. Predict the wavelength of the transition from the 4D3/2 level to the 2P3/2 level of the ground state term.
Figure 7.6 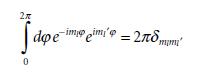
Transcribed Image Text:
2x 0 doe-imimi' = 278mm 2πδ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To calculate the constant of spinorbit coupling A for the initial condition 2 2P and feeling of exci...View the full answer

Answered By

Hassan Ali
I am an electrical engineer with Master in Management (Engineering). I have been teaching for more than 10years and still helping a a lot of students online and in person. In addition to that, I not only have theoretical experience but also have practical experience by working on different managerial positions in different companies. Now I am running my own company successfully which I launched in 2019. I can provide complete guidance in the following fields. System engineering management, research and lab reports, power transmission, utilisation and distribution, generators and motors, organizational behaviour, essay writing, general management, digital system design, control system, business and leadership.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The 2 P 2 S transition of Li is split by 0.34 cm 1 . Find spin orbit coupling constant for the 2P multiplet and compare to the values found in the previous problem for Ar + . Data from previous...
-
Consider the loss function of linear regression given by: J(8,, 0,). Given (6,, e,) = (0, 0.5), Estimate %3D using the data points below: ae, 4. 7.0 8.0 10.0 2.5 3.5 5.5
-
Given the following energy level diagram for an atom that contains an electron in the n = 3 level, answer the following questions. a. Which transition of the electron will emit light of the lowest...
-
Write a program that plots average path length versus the number of random edges as random shortcuts are added to a 2-ring graph on 1,000 vertices.
-
Find the time the earth takes to orbit the sun.
-
Identify some pathogen-associated molecular patterns (PAMPs) that are recognized by pattern recognition receptors (PRRs). What is the significance of the interactions between these molecules?
-
An object moves with constant velocity. What can you say about the work done on a system that includes only this object? \(\cdot\)\(\cdot\)
-
Ajax Cleaning Products is a medium-sized firm operating in an industry dominated by one large firmTile King. Ajax produces a multiheaded tunnel wall scrubber that is similar to a model produced by...
-
1. A scientific committee is to be formed form 6 Indians and 8 foreigners, which includes at least 2 Indians and double the number of foreigners as Indians. Then the number of ways the committee and...
-
This problem illustrates the separation of internal and external motions for a one-dimensional rigid rotor. Consider a diatomic molecule consisting of masses m 1 and m 2 at positions x 1 and x 2 ....
-
Are two-electron transitions permitted by electric dipole selection rules? To arrive at your answer, first consider the wavefunction to be a simple product of spinorbitals. (This is called a Hartree...
-
Look up the standard enthalpies of formation and the entropies of solid ice and liquid water and verify that ice may spontaneously melt at NTP (20 C, 1 atm).
-
What is the profit/loss on a 1-Year long forward EUR at t=1 when X1USD/EUR = 1.28; F1USD/EUR = 1.22 and the size of the contract is EUR 350,000? Assume you want a short position in AUD in a 1-Year...
-
Light Up Ltd. ("Light Up") is a solar panel manufacturer listed on the main board of the Johannesburg Stock Exchange (JSE) in the energy sector. The entity was established in April 2019 and has a 30...
-
What is meant by the "revolving door" between business and government regulatory bodies? Why do many see this as a problem?
-
The table below displays information from a recent year for Republica, and the U.S. Country United States Population Size 325 million Number of Eggs Consumed 5.070 billion Number of Cattle 75.9...
-
Question 2 - Market Dominance (1% of your final grade) Your firm has conducted some market research on the major soda manufacturers in the market: Coke, Pepsi, and Sprite. Based upon this market...
-
Jane wants to estimate the proportion of students on her campus who eat cauli flower. After surveying 20 students, she nds 2 who eat cauliflower.Obtain and interpret a 95% condence interval for the...
-
Ask students to outline the reasons why the various elements of culture (social structures and control systems, language and aesthetics, religion and other belief systems, educational systems, etc.)...
-
Draw all constitutional isomers with molecular formula C 5 H 10 that possess one p bond.
-
Rank the following anions in terms of increasing basicity:
-
Identify the alkene that would yield the following products via ozonolysis: a. b. c. d.
-
Rewrite this AI: This piece offers important perspectives on the elements that affect the choice to pursue AACSB accounting accreditation. It underscores the intricate interaction between perceived...
-
Please answer the questions and SHOW YOUR WORK. Mel Co. Balance sheet - Before Investment at 12/31/23 Mel Co. Balance Sheet - Before Investment 12/31/2023 Cash 5,000 ...
-
Which is not true about the new auditing standards on estimates and evidence? The PCAOB has issued a new standard on estimates and fair values that is already in effect. The ASB is currently...

Study smarter with the SolutionInn App


