The half-life of ({ }^{239} mathrm{Pu}) is 24,110 years. Find its mean-life. (Delta=M-A) is the mass excess,
Question:
The half-life of \({ }^{239} \mathrm{Pu}\) is 24,110 years. Find its mean-life.
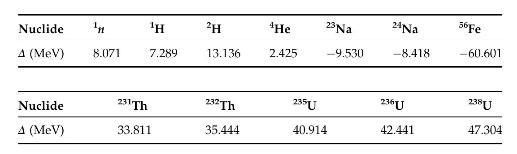
\(\Delta=M-A\) is the "mass excess," where \(M\) is the mass of a nuclide and \(A\) its mass number. These data are given in the Nuclear Wallet cards, and elsewhere. The data given below may be useful in solving some of the problems.
Transcribed Image Text:
Nuclide n H 2H 4He 23 Na 24Na A (MeV) 8.071 7.289 13.136 2.425 -9.530 -8.418 56Fe -60.601 Nuclide 231 Th 232 Th 235U 236U 238U A (MeV) 33.811 35.444 40.914 42.441 47.304
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Match the following mental health practitioners with the appropriate description. 1. Psychiatrist 2. Clinical psychologist 3. Counseling psychologist 4. Psychoanalyst a. PhD specializing in the...
-
Density of natural boron is \(2.4 \mathrm{~g} / \mathrm{cc}\). Given: \(\sigma_{a}\) of \({ }^{10} \mathrm{~B}=4000 \mathrm{~b}\), and \(\sigma_{a}\) of \({ }^{11} \mathrm{~B}=0 \mathrm{~b}\). If the...
-
If the initial energy of the neutron is \(2 \mathrm{MeV}\), find the maximum energy loss to the elastically scattered neutron in \({ }^{2} \mathrm{H},{ }^{56} \mathrm{Fe}\), and \({...
-
The following is information for a perfectly price discriminating monopolist. Demand: P = 65 0.02Q Marginal revenue = P = 65 0.04Q Marginal cost = ATC = 4 Calculate the producer surplus for the...
-
The Fed creates $100,000 in new money that is deposited in someone's checking account in a bank. What is the maximum change in the money supply if the required reserve ratio is 5 percent, 10 percent,...
-
Light is incident on a grating at an angle ? as shown in Figure show that bright fringes occur at angles ? that satisfy the equation d(sin ? + sin ?) = m?, for m = 0, 1, 2, . . . (compare this...
-
Design Data sold a piece of machinery to HHB Drafting Company. However, after HHB had taken possession of the machine, it discovered damage and revoked the contract. The court found that the...
-
Payroll Tax Entries Below is a payroll sheet for Otis Import Company for the month of September 2010. The company is allowed a 1% unemployment compensation rate by the state; the federal unemployment...
-
Map the EER model of the following databases, to a relational model. Show all integrity types(Clarify your answer by underline the Primary key, and dotted line the Foreign Key). (a) International...
-
Find the \(Q\)-value of the reaction: \(\mathrm{Na}^{23}+\mathrm{n}^{1} ightarrow \mathrm{Na}^{24}+\gamma\). Assume kinetic energy of the incident neutron to be negligible. \(\Delta=M-A\) is the...
-
Calculate the energy of the \(\alpha\) particle emitted by \({ }^{235} \mathrm{U}\). \(\Delta=M-A\) is the "mass excess," where \(M\) is the mass of a nuclide and \(A\) its mass number. These data...
-
A random sample of 500 households was identified in a major North American city using the municipal voter registration list. Five hundred questionnaires went out, directed at one adult in each...
-
VIETNAM'S business model and its influence on global market. TOPIC - Business control, Financial Requirements, Forecasting and Financial Data. With references and pls avoid repetition of lines.
-
Based on the pet food industry in Canada, provide a detailed analysis on the nature of competiton and competitor's analysis for an upcoming pet food brand based on the following factors: 1. Who are...
-
On January 1, Year 1, Chester Co. began construction of a small building. The following expenditures were incurred for construction in Year 1: January 1 $125,000 May 1 $104,000 Oct 1 $124,000...
-
What part of the Modified Logic Model would you be referring to if you noted that even though it cost $20 per member of the community to get the flu vaccine from your campaign, the cost of every...
-
Air saturated with chlorobenzene at 113 C and 101.3 kPa is cooled to 58.3 C at constant pressure. Estimate the percentage of the chlorobenzene originally in the vapor that condenses. Percent...
-
(a) What are the magnitude and direction of the magnetic field a distance a below the current sheet? (b) What are the magnitude and direction of the magnetic field a distance a above the current...
-
The percentage of completion and completed contract methods are described in the FASB ASC. Search the codification to find the paragraphs covering these topics, cite them, and copy the results.
-
Compare the following three isomeric dienes: (a) Which compound will liberate the least heat upon hydrogenation with 2 mol of hydrogen gas? Why? (b) Which compound will liberate the most heat upon...
-
Identify the most stable compound:
-
Draw an energy diagram showing the relative energy levels of the MOs for 1,3,5,7- octatetraene and identify the HOMO and LUMO for both the ground state and the excited state.
-
Prove that the ideal gas temperature scale as defined by its equation of state is identical to the thermodynamic temperature scale as defined by the second law
-
The shaft rotating at 750 rpm carries a spur gear B with 79 teeth and a module of 5 mm. The teeth are 20, full-depth, involute form. The gear receives 25 kW from a pinion directly above it as shown....
-
9. (3) Assuming a fin with insulated tip, calculate the heat transfer coefficient in W/m2.K.

Study smarter with the SolutionInn App


