(a) Write reactions to show how you could convert 2-methyl-2-butene into 2-methyl-1,3-butadiene. (b) Write reactions to show...
Question:
(a) Write reactions to show how you could convert 2-methyl-2-butene into 2-methyl-1,3-butadiene.
(b) Write reactions to show how you could convert ethylbenzene into the following compound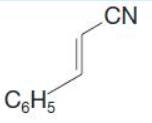
(c) Write structures for the various Diels–Alder adduct(s) that could result in reaction of 2-methyl-1,3-butadiene with the compound shown in part (b).
Transcribed Image Text:
C6H5 CN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
a The reaction involves an intramolecular addition of ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Write reactions to show how nitric and sulfuric acids are produced in the atmosphere. Write reactions to show how the nitric and sulfuric acids in acid rain react with marble and limestone. (Both...
-
Show how you could convert the ethyl ester of Z-Phe-Gly to Leu-Phe-Gly (as its ethyl ester) by the active ester method.
-
Show how you could prepare each of the following compounds. Use the starting material indicated along with ethyl acetoacetate or diethyl malonate and any necessary inorganic reagents. Assume also...
-
Q8 Question: 9 A small particle of mass m moving inside a heavy, hollow and straight tube along the tube axis undergoes elastic collision at two ends. The tube has no friction and it is closed at one...
-
3(x 2y) = 9 Determine the slope and the y-intercept. Use the slope and the y-intercept to graph the equation by hand.
-
From what country(ies) is(are) A-Zs victims?
-
Find the percentile that corresponds to an age of 47 years old. Use the data set, which represents the ages of 30 executives. 43 57 65 47 57 41 56 53 61 54 56 50 66 56 50 61 47 40 50 43 54 41 48 45...
-
A cement manufacturer has supplied the following data: Tons of cement produced and sold 220,000 Sales revenue ...........$924,000 Variable manufacturing expense ...$297,000 Fixed manufacturing...
-
Date March 1 March 5 March 9 March 18 March 25 March 29 Activities Beginning inventory Purchase Sales Purchase Purchase Sales Totals Units Acquired at Cost $52.60 per unit 180 units 265 units 125...
-
Binary tree can be constructed back using: a) Pre-order sequence and in-order sequence b) Pre order sequence and post order sequence c) Post order sequence and in-order sequence d) a or c
-
Predict the products of the following reactions. (a) (b) HCI
-
Elucidate the structure of the compound that gives the following spectroscopic data. Assign the data to specific aspects of your proposed structure. MS (m/z); 120, 105 (base peak), 77 1 H NMR ():...
-
Should George Keyworth have been asked to resign? Why or why not?
-
Consider the simple regression model Y =B + Bx +4 t = 1,2, ...,T where u, pu, + & with lpl < 1 and E(e) = 0, E(?) = o?, E(EE) = 0,t #s a. What are the consequences of having first order...
-
196. By which process man has obtained many breeds of plants and animals for agriculture, horticulture, food and security? (1) Natural selection (2) Random breeding (3) Artificial selection (4) Both...
-
An m = 0.5 kg compresses a spring of force constant 500 N/m a distance 0.2 m from its natural length. When the block is released, it travels up a rough incline that is h = 0.4 m high and 2 m long....
-
Calculate the investment and operation cost of all three alternatives according to the following loading profile over the first 10 years. Assume that energy costs 30 Eur / MWh and the total running...
-
A 20-Kilogram car traveling east at 6 meters per second collides with a 30-Kilogram car that is traveling west. If both the cars come to rest immediately after the collision, what was the speed of...
-
Theorem 3.8 assumes that all calculations for det(A) are done by exact arithmetic. As noted previously, this is usually not the case in software. Hence, computationally, the determinant may not be a...
-
Review Exhibit 11.4. Analyze each product on the graph according to the characteristics that influence the rate of adoption. For example, what can you conclude from the data about the relative...
-
Suggest a mechanism for thisreaction: CH3 CH, CH3 CH3 N. CHCI CI
-
In addition to the reaction shown on p. 353, Diphenhydramine can also be prepared by heating bromo diphenyl methane and 2-(dimethyl lamino)-1-ethanol in a polar solvent. Show a mechanism for...
-
Another Diphenhydramine synthesis is shown in the following equation: (a) Show a mechanism for the first step in this synthesis. (b) Explain which mechanism is occurring in the secondstep. OCH CH...
-
A project that costs $4,450 to install will provide annual cash flows of $1,500 for each of the next 6 years. a. What is NPV if the discount rate is 11%? b. How high can the discount rate be before...
-
Prove MM1: Both Umbrella Inc. and Prixxa Inc. produce $300,000 of cash flow each year. Umbrella has no debt outstanding, and its cost of equity capital is 14 percent. But Prixxa has 1,000,000 depts...
-
How did they find the over head rate percentages for current year and last year? A job order cost sheet for Cullumber Company is shown below. Job No. 92 For 2,000 Units Date Direct Materials Direct...

Study smarter with the SolutionInn App


