In addition to the reaction shown on p. 353, Diphenhydramine can also be prepared by heating bromo
Question:
In addition to the reaction shown on p. 353, Diphenhydramine can also be prepared by heating bromo diphenyl methane and 2-(dimethyl lamino)-1-ethanol in a polar solvent. Show a mechanism for thisreaction:
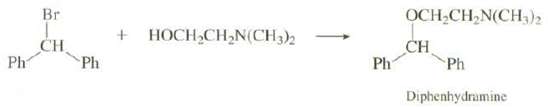
Transcribed Image Text:
Br OCH,CH,N(CH,)2 HOCH CH,N(CH3)2 CH Ph CH Ph Ph Ph Diphenhydramine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 53% (15 reviews)
Ph Br CH Ph SN1 ...View the full answer

Answered By

Issa Shikuku
I have vast experience of four years in academic and content writing with quality understanding of APA, MLA, Harvard and Chicago formats. I am a dedicated tutor willing to hep prepare outlines, drafts or find sources in every way possible. I strive to make sure my clients follow assignment instructions and meet the rubric criteria by undertaking extensive research to develop perfect drafts and outlines. I do this by ensuring that i am always punctual and deliver quality work.
5.00+
6+ Reviews
13+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Diphenhydramine can also be synthesized by heating bromo diphenyl methane with the amino alcohol shown here. Offer a reason why the oxygen, rather than the nitrogen, of this compound acts as the...
-
In addition to the FASB's statement on accounting for research and development activities, the EITF has addressed three implementation issues. List and briefly summarize each of these issues.
-
In addition to the proton marked Ha in m-nitrostyrene in Figure 13.18, there are two other vinylic protons. Assuming that the coupling constant between the two geminal protons in ArCHoeCH2 is 2 Hz...
-
What is the type of the expressions computed on these two lines? 4 > 5 print (4>5)
-
For each of the costs listed below, indicate whether it is: (a) A product or period cost (b) A variable or fixed cost (c) A manufacturing or non-manufacturing cost. 1. Advertising costs of Nike. 2....
-
Two firms, Sludge Oil and Northwest Lumber, have access to five production processes, each one of which has a different cost and gives off a different amount of pollution. The daily costs of the...
-
Which of the following statements about lift is false? a. Lift can be interpreted as how much more likely the antecedent and consequent itemsets occur together than if they were totally unrelated. b....
-
Carson Trucking is considering whether to expand its regional service center in Moab, Utah. The expansion requires the expenditure of $10,000,000 on new service equipment and would generate annual...
-
The functions fand g are defined as follows. f(x)=x-6x-4 and g(x)=- x-4 3x-5 Find f(x+8) and g (4). Write your answers without parentheses and simplify them as much as possible. f(x+8)= *(4)-0 g = H 5
-
Determine the Miller indices for the planes shown in the following unitcell: +y B.
-
Suggest a mechanism for thisreaction: CH3 CH, CH3 CH3 N. CHCI CI
-
Another Diphenhydramine synthesis is shown in the following equation: (a) Show a mechanism for the first step in this synthesis. (b) Explain which mechanism is occurring in the secondstep. OCH CH...
-
In Problems 1322, for the given functions f and g, find: (a) (f g)(4) (b) (g f)(2) (c) (f f)(1) (d) (g g)(0) f(x) = 3 x+1' g(x) = x
-
Past data indicate that the variance of measurements made on sheet metal stampings by experienced quality-control inspectors is 0.18 (inch) \({ }^{2}\). Such measurements made by an inexperienced...
-
What is globalization?
-
Search online for information about microfinance in developing countries. What are the potential benefits? What criticism has it received?
-
Comment on the following statement: Globalization is an event of the post-1980s. Prior to this time, we never had to worry about globalization and its effects.
-
a. Determine the price elasticity of demand at each quantity demanded using the arc or midpoint formula: Percentage change in quantity demanded = (Q 2 - Q 1 )/Q 1 divided by percentage change in...
-
We have \(N=396\) observations on employment at fast-food restaurants in two neighboring states, New Jersey and Pennsylvania. In Pennsylvania, the control group \(d_{i}=0\), there is no minimum wage...
-
Show that the peak of the black body spectrum as a function of ? is given by eq. (22.14) kg T Wmax = 2.82
-
Solve each system by graphing. 2.x +3y = 6 4.x + 6.y = 12 -4 -3 in # 3 10 + || + 15 fet en - HAT
-
Draw an energy diagram for the three molecular orbitals of the cyclopropenyl system (C3H3). How are these three molecular orbitals occupied in the cyclopropenyl anion, cation, and radical? Which of...
-
Cyclopropanone is highly reactive because of its large amount of angle strain, but methylcyclopropenone, although even more strained than Cyclopropanone, is nevertheless quite stable and can even be...
-
Cycloheptatrienone is stable, but cyclopentadienone is so reactive that it can?t be isolated. Explain, taking the polarity of the carbonyl group into account. Cycloheptatrienone Cyclopentadienone
-
The highest WACC is 11%, the baseline WACC is 7.4% and lowest WACCS is 2.8%. To derive a WACC that better accounts for extreme scenarios, you decide to assign the following probability weights: High-...
-
Give 2 examples of the ff: Statutory law Regulatory law Common law Include the title of each example and explain why you chose these examples ( 1 to 2 sentences ) . Note: Write your reference ( s )
-
The Adams family includes a financially well-informed couple, both aged 36, and two children aged 4 and 6. The family is financially sound but suffered badly during the tech meltdown in 2000. The...

Study smarter with the SolutionInn App


