Deduce the structures of compounds AD. Draw structures that show stereochemistry where appropriate: CgH1,04 D OH (optically
Question:
Deduce the structures of compounds A–D. Draw structures that show stereochemistry where appropriate: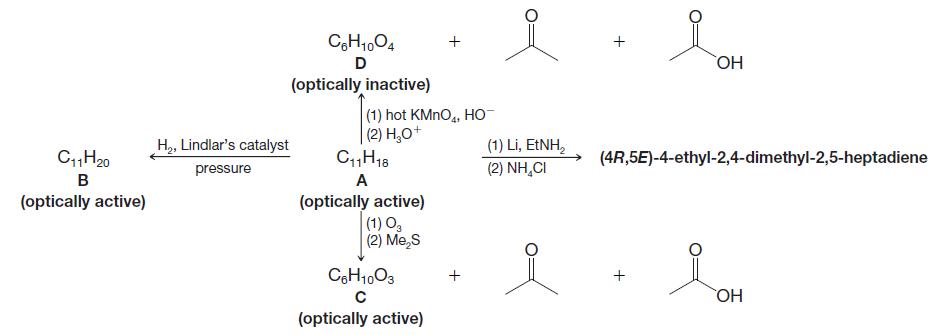
Transcribed Image Text:
CgH1,04 D OH (optically inactive) (1) hot KMNO,, HO | (2) H,0+ CH18 (1) Li, EINH, (2) NH,CI H2, Lindlar's catalyst C,H20 (4R,5E)-4-ethyl-2,4-dimethyl-2,5-heptadiene pressure в A (optically active) (optically active) (1) O, (2) Me,S CoH1003 HO (optically active)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)

Answered By

Carlos otieno
I am readily available to provide correct answers to students on time.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Deduce the structures of the following compounds. (a) C4H8O: IR 1717 cm-1 NMR 0.95 (3H, t, J = 8 Hz); 2.03 (3H, s): 2.38 (2H. q, J = 8 Hz) (b) A compound with molecular mass = 70.1, IR absorption...
-
Given the following information, elucidate the structures of compounds A and B. Both compounds are soluble in dilute aqueous HCl, and both have the same molecular formula. The mass spectra of A and B...
-
Determine the structures of compounds A through G, including stereochemistry where appropriate. C6H10 H,, Pt CH H,so, (2) H,o* heat KMnO4 I warm, concd H,so, heat C10H 16 CH ) CH3Mgl (2) H,o* H so...
-
The General Auditors Office (GAO) of ABC jurisdiction issued a report on the XYZ Electric Cooperative, a large member-owned utility. This report reviewed the work of MNO Consulting. MNO found...
-
What roles do specialization and division of labor play in economists' support of free trade?
-
Discuss the five paradigms of quality.
-
We will see in Chapter 8 that the pressure drop in fully developed pipe flow is sometimes computed with the aid of a friction factor, defined by \[ f=\frac{\Delta p}{\frac{1}{2} ho V^{2}}...
-
Janet and James purchased their personal residence 15 years ago for $300,000. For the current year, they have an $80,000 first mortgage on their home, on which they paid $5,600 in interest. They also...
-
What do you think is important to employees to make them stay and continue with a company through all the growth and changes that can happen?
-
A trader takes a short position in a 5 Eurodollar futures contract at the futures price of 98.14. A short time later, the trader closes out the position at a price of 98.27. What is the traders total...
-
Write a reasonable and detailed mechanism for the following transformation: concd H,SO, + H,O heat H
-
In one industrial synthesis of ethanol, ethene first undergoes an addition reaction with sulfuric acid, and this product undergoes hydrolysis to ethanol. Write a mechanism for the addition of...
-
Experiments on learning in animals sometimes measure how long it takes mice to find their way through a maze. The mean time is 18 seconds for one particular maze. A researcher thinks that a loud...
-
What are the specified tolerances that perishable stock should remain within?
-
Define the purpose of "Access Vector" and "Privileges Required" CVSS metric.
-
What is the allowable depreciation for the computer in year one?
-
What Division and Section would you find information on brick masonry?
-
Describe a debit, credit , T -account, asset,liability, equity, revenue, and expense.
-
Describe the structure of hyaline cartilage. Where can it be found in the body?
-
Answer the following two independent questions. a. MM Corporation is considering several proposed investments for the coming budget year. MM produces electrical apparatus for industrial complexes....
-
Show the conjugate bases of these species: a) H-0-H_bH-0 H CH-N-H d) H-C-C-H H
-
Complete these acid-base equations. Use the curved arrow method to show the electron movement in the reactions. Base Acid a) NH, + H: b) CHO + HO: Conjugate acid Conjugate base
-
Indicate whether each of these species is a Lewis acid, a Lewis base, or both: H 1. a) H-C I H d) CH3-N-H T H b) H-O-H :8-3-6: :ci: e) :CI-AI :CI: H c) H-B 1 H
-
The market price of a semi-annual pay bond is $957.95. It has 15.00 years to maturity and a coupon rate of 7.00%. Par value is $1,000. What is the yield to maturity? The market price of a semi-annual...
-
Compute the accumulated sum of the following payments: (a) $400 every six months for 4 years at 8% compounded semi-annually (b) $250 per month for 5 years at 7.5% compounded monthly.
-
The output is wrong. Sometimes a program lacking input will produce wrong output (as in this case), or no output. Remember to always pre-enter needed input. Type 2 in the input box, then click "Run...

Study smarter with the SolutionInn App


