Deduce the structures of compounds EL in the roadmap below. E (CH13 Br) HBr (no peroxides) F
Question:
Deduce the structures of compounds E–L in the roadmap below.
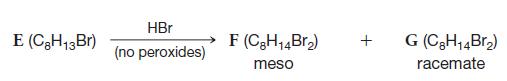
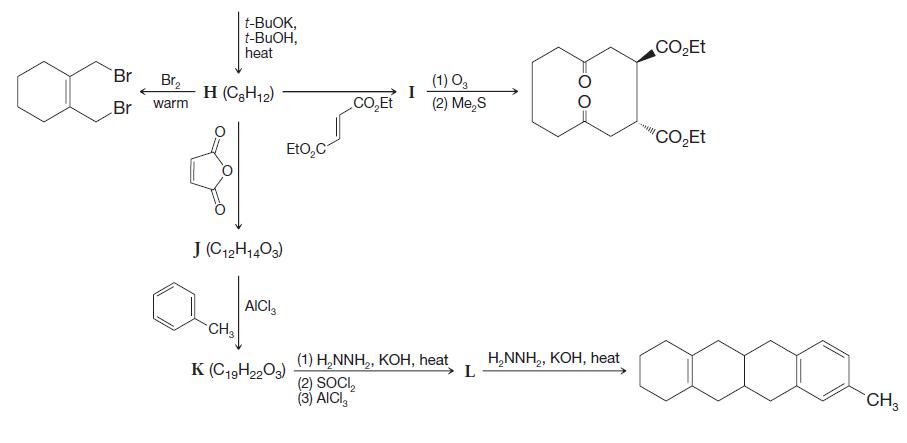
Transcribed Image Text:
E (C₂H13 Br) HBr (no peroxides) F (C8H₁4Br₂) meso + G (C₂H₁4Br₂) racemate
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
Answer Compound E C6H14 Compo...View the full answer

Answered By

Justin Akengo
I am writing in application for the tutor position with your organisation. I am experienced in tutoring students of all abilities and I believe I am the ideal candidate for this position.
I work with students of all ages, from elementary school to college level. Whether the subject is science, Mathematics or basic study skills, I break material down into easy-to-understand concepts. In your job posting, you asked for someone who can tutor in a variety of subjects. I am comfortable explaining calculus to a college student or working with a kindergartener on spelling fundamentals.
Below are just a few core skills and qualifications I posses as a tutor;
Adept at creating study materials in a variety of academic subjects to help students improve their test scores and GPAs.
Strong interpersonal skills in working with students to help them achieve and succeed.
Have written study books adopted by a high school and a college to help students improve their skills in English and mathematics.
Have won several “Tutor of the Year” awards for work with high school and college students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Using the information provided below, deduce the structures of compounds A, B, C, and D: 1) EtMgBr 1) O3 (C10H12) (C,H160) 2) H,0 2) DMS (C9H100) [H*), (CH3)2NH AICI3 (-H20)
-
Deduce the structures of the following compounds. (a) C4H8O: IR 1717 cm-1 NMR 0.95 (3H, t, J = 8 Hz); 2.03 (3H, s): 2.38 (2H. q, J = 8 Hz) (b) A compound with molecular mass = 70.1, IR absorption...
-
Deduce the structures of compounds AD. Draw structures that show stereochemistry where appropriate: CgH1,04 D OH (optically inactive) (1) hot KMNO,, HO | (2) H,0+ CH18 (1) Li, EINH, (2) NH,CI H2,...
-
There is a crop that the value p next month is random. The value can be either small p=1 or large P=3 depending on the weather. Half of people are optimists believing the value of the crop will be...
-
6p + 2 /8 4p 1 /6 Describe the solution set as an inequality, in interval notation, and on a graph.
-
Alexander Corporation reports the following components of stockholders' equity on December 31, 2015: Common stock-$25 par value, 50,000 shares authorized, 30,000 shares issued and outstanding . . . ....
-
Discuss the use of EVM for project portfolio analysis.
-
Notes Receivable Patterson Company is a large diversified business with a unit that sells commercial real estate. As a company, Patterson has been profitable in recent years with the exception of the...
-
At Kenneth's company, two types of candles are created from the common melting and molding process: pillar candles and votive candles (eight votives per pack; one pack is one unit). The costs of the...
-
Lindsay Kay approached her friend Michael Wait about going into business together as co owners of a gardening centre. Lindsay loved gardening and had some skills as an accountant. Michael was a...
-
Synthesize 2-chloro-4-nitrobenzoic acid from toluene and any other reagents necessary. Begin by writing a retrosynthetic analysis.
-
Elucidate the structures of compounds A through I in the following road map problem. Specify any missing reagents. A (C5H) Br., warm (1 molar equiv.) F CH,ONA (2 molar equiv.) G 1 HBr (no ROOR) H...
-
a) What did Soldau do that was wrong? b) How did the court rule in favor of Organon? c) But Soldau got the letter back - doesn't that mean the mailbox rule doesn't apply?
-
Verizox Company uses a job order cost system with manufacturing overhead applied to products based on direct labor hours. At the beginning of the most recent year, the company estimated its...
-
Oslo Company prepared the following contribution format income statement based on a sales volume of 1,000 units (the relevant range of production is 500 units to 1,500 units): Sales Variable expenses...
-
Melissa recently paid $640 for round-trip airfare to San Francisco to attend a business conference for three days. Melissa also paid the following expenses: $635 fee to register for the conference,...
-
The Cheyenne Hotel in Big Sky, Montana, has accumulated records of the total electrical costs of the hotel and the number of occupancy-days over the last year. An occupancy-day represents a room...
-
A company constructs a building for its own use. Construction began on January 1 and ended on December 30. The expenditures for construction were as follows: January 1, $660,000; March 31, $760,000;...
-
Write an application that displays a box, an oval, an arrow and a diamond using asterisks (*), as follows: Discuss.
-
Pearson Education, a publisher of college textbooks, would like to know if students prefer traditional textbooks or digital textbooks. A random sample of students was asked their preference and the...
-
Draw both chair conformations of 1-methyl-1-phenylcyclohexane. Which is more stable by how much energy?
-
Explain whether the methyl is axial or equatorial in this compound: CH3 H C-C-CH3 CH3
-
Draw both chair conformations for menthol (a component of peppermint oil) and its stereo isomer, neo menthol. Which groups are axial and which groups are equatorial? Explain which conformation is...
-
A positive charge q = +8 nC is at the origin, and a second positive charge q = +12 nC is on the x axis at a = 4 m, Find the net electric field (a) at point P, on the x axis at x=7 m, and (b) at point...
-
8. Hooten Carpentry had the following accounts and account balances after adjusting entries. Assume all accounts have normal balances. Prepare the adjusted trial balance for Hooten Carpentry as of...
-
Explain at least one major difference that exists today between US GAAP and IFRS on the accounting for foreign currency transactions. Be specific.

Study smarter with the SolutionInn App


