Use a table of p K a values, such as Table 3.1, to estimate pKa values for
Question:
Use a table of pKa values, such as Table 3.1, to estimate pKa values for any potentially acidic hydrogens in each of the species you listed in part 1 (or for the conjugate acid).
Table 3.1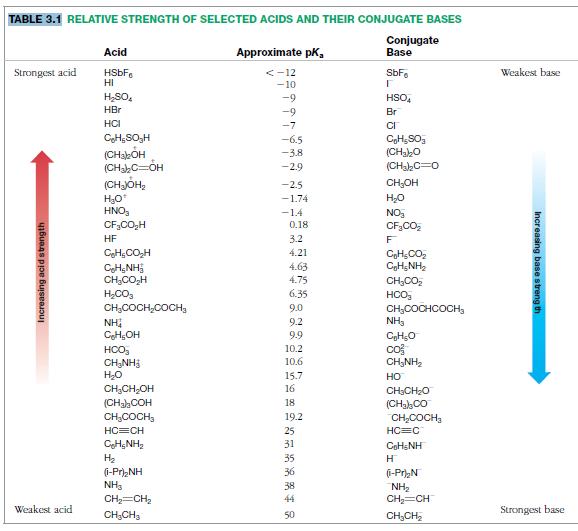
Transcribed Image Text:
TABLE 3.1 RELATIVE STRENGTH OF SELECTED ACIDS AND THEIR CONJUGATE BASES Conjugate Base Acid Approximate pk, Strongest acid <-12 -10 SbF. Weakest base HI HSO, -9 HSO, HBr -9 Br HCI -7 CH;SO,H -6.5 (CHabÓH (CHC=OH (CHJÓH2 H30 HNO, -3.8 (CHakO -29 (CHahC=0 -2.5 CH;OH -1.74 -14 0,18 NO CF3CO, CF,CO,H HF 3.2 4.21 CH,CO, CH;NH2 4.63 CH,NH CH,CO,H 4.75 CH,C0, H;CO, 6.35 HCO, CH,COCH,COCH, 9.0 9.2 CH;COCHCOCH, NH3 NH CH,OH 9.9 HCO, CH;NH3 H20 10.2 10.6 co CH,NH2 15.7 16 но CH,CH,OH CH3CH2O (CHaCOH 18 (CHakc0 CH,COCH, 19.2 CH,COCH, HC=CH 25 HC=C CH;NH, H2 31 COHSNH 35 36 (1-Pr),NH NH3 (1-Pr),N 38 NH2 CH=CH CH=CH 44 Weakest acid Strongest base CH,CH3 50 CH,CH2 Increasing acid strength Increasing base streng th
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)

Answered By

SAKET KUMAR
Currently I'm a chemical engineering student at Visvesvaraya National Institute Of Technology, Nagpur, India. I'm working in this field from last 2 years. I love to solve and clear the doubt of students as simple as possible.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Question Posted:
Students also viewed these Sciences questions
-
Use a table of values to estimate the value of the limit. If you have a graphing device, use it to confirm your result graphically. lim
-
Use a table of values to estimate the value of the limit. If you have a graphing device, use it to confirm your result graphically. Vx +4 19. lim tan 3x 20. lim -o tan 5x -2 9* - 5* 21. lim 22. lim
-
Use a table of increasing values of x to find each of the following limits. If no limit exists, say the limit is undefined. a. b. c. 9x-1 lim f(x) if f(x) = 3x - 5 lim g(x) ifg(x) = 3rth 4x +1 lim...
-
Factor each polynomial. 64y 9 + z 6
-
How is the "fallacy of composition" related to the difference between micro demand and supply curves and macro aggregate demand and supply curves?
-
I. Which of the following is correct for a load instruction? Refer to Figure 4.10. a.MemtoReg should be set to cause the data from memory to be sent to the register file. b.MemtoReg should be set to...
-
Water flows through the curved hose shown in Fig. P4.49 with an increasing speed of \(V=10 t \mathrm{ft} / \mathrm{s}\), where \(t\) is in seconds. For \(t=2 \mathrm{~s}\) determine (a) the component...
-
The capital accounts of Jonathan Faber and Faheem Ahmad have balances of $150,000 and $110,000, respectively. Lauren Wells and Rachel Lee are to be admitted to the partnership. Wells buys one-fifth...
-
Harris Fabrics computes its plantwide predetermined overhead rate annually on the basis of direct labor-hours. At the beginning of the year, it estimated that 29,000 direct labor-hours would be...
-
Richard chooses technique 0 and 2 requiring 10+10-20 efforts and provising 10+11=21 benefits. Hence, 21 is returned as the output Example 2: input1: 3 input2: (10,10,10,10) input3: (10,11,12,15)...
-
Predict the outcome of the following reaction. NH,
-
List the acids you gave as answers to Problem 3.22 in order of decreasing acidity. Problem 3.22 What is the conjugate acid of each of the following bases? (a) HSO 4 (b) H 2 O (c) CH 3 NH 2 (d) NH 2...
-
Define the adiabatic saturation temperature.
-
DSI109 course . Considering your own data, why might you pivot or group that data?
-
Economies of scale can be defined as the connection between the size of the corporation as well as that the corporation's capability to sell its goods as well as its products minimal potential cost....
-
Find the new car prices for two popular cars: Honda Accord and the Ford Fusion. What are their prices?Are the two cars closely priced? Do you think physically similar goods are close substitutes when...
-
Question 2. Suppose that, in an election with two candidates, there are six voters, 1, 2, 3, 4, 5, and 6, who are all concerned with the same issue. Voter i has a single-peaked preference with a peak...
-
Michael decides to drive because he's excited about his new car, plus, he thinks the firm owes him for having suffered too many poor flights on previous trips. What is the opportunity cost to the...
-
Labe this diagram of a human eye. ab G
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
Explain whether you would expect KBr or CH3Br to have the higher melting point.
-
Which of these isomers would you expect to have the higher boiling point: Explain? CH 3 CH 2 CH 2 OH or CH 3 CH 2 OCH 3
-
Which of these compounds would you expect to be more soluble in water? Explain? CHCHCHCHCOH or CH3CHCHCHCHCOH
-
Salmon ASA has just issued a callable seven-year, 8% coupon bond with coupon payable annually. The bond can be called at par in two years or anytime thereafter on a coupon payment date. It has a...
-
Lamda corporation wants to acquire another company within its industry for $100m and it expects the acquisition to contribute to its free cash flow by $5m the first year, and this contribution is...
-
Dewan INC. has several divisions, each with a manager responsible for the operations of the division. Each division of Dewan controls product design, sales, pricing, operating costs, and profits.....

Study smarter with the SolutionInn App


