Classify the following monosaccharides: CH,OH C=0 HC=0 HC-O -H OH - - H-
Question:
Classify the following monosaccharides:
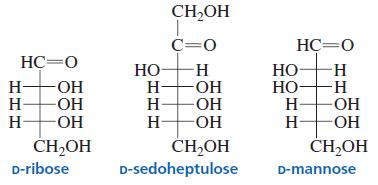
Transcribed Image Text:
CH,OH C=0 HC=0 HC-O НО -H Н— ОН ОН OH Но- Но- H- H- -H- -- OH ОН H-OH -ОН H- -O- ČH,OH ČH,OH D-sedoheptulose ČH,OH D-ribose D-mannose HH HHH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Classify each of the following monosaccharide?s: la) 0 (c) (b) (d) CH- . H- C=0 C=0 -- -- -- -- -- -- -- -- -- H2OH -- CH- Ribulose CH Threose CH- Tagatose 2-Deoxyribose
-
Classify the following monosaccharides. (Examples: D-aldohexose, L-ketotetrose.) (a) (+)-glucose (b) (-)-arabinose (c) L-fructose (d) (e) (f) (g) CHO HO HO OH HO CH,OH +)-gulose CH,OH HOH CH,OH...
-
Classify each of the following monosaccharides as either d or l, as either an aldo or a keto sugar, and as a tetrose, pentose, or hexose: (a) (b) (c) (d) (e) . C=0 . - - CH- 12 . - - - - - H,OH
-
A survey of U.S. adults found that 33% name professional football as their favorite sport. You randomly select 14 U.S. adults and ask them to name their favorite sport. Find the probability that the...
-
You have been sent to India to investigate the occurrence of goiter disease attributed to iodine deficiency. As part of your investigation, you must make field measurements of traces of iodide ( I- )...
-
Three brands of batteries are under study. It is suspected that the lives (in weeks) of the three brands are different. Five batteries of each brand are tested with the following results: (a) Are the...
-
What is a contra-asset? Give an example of one.
-
CLV calculation example Using the Tier One customer data in the scenario, the CLV for a Tier One customer is calculated as follows: Where Tier One has GP = $4,500 | RR = 0.85 | DR = 0.10| AC = $2,500...
-
3. The market share of Seattle Mills is $407 million. The claims on Seattle Mills are split between common stock and a 12% debt that has a face value of $350 million and that matures in five months....
-
A new police car costs the Bay City Police Department $26,000. The annual maintenance cost for a car depends on the age of the car at the beginning of the year. (All cars accumulate approximately the...
-
Indicate if each of the following items would be recognized in TelCan Ltd.s financial statements for 20X3 and, if so, what elements would be recognized. For any items that would not be recognized,...
-
a. Are D-erythrose and L-erythrose enantiomers or diastereomers? b. Are L-erythrose and L-threose enantiomers or diastereomers?
-
Thermal neutrons fall normally on the surface of a thin gold foil consisting of stable Au197 nuclide. The neutron flux density is J = 1.0 1010 part./(s cm2). The mass of the foil is m = l0 mg. The...
-
Distinguish between auditors report and auditors certificate.
-
State the powers of an auditor of an insurance company.
-
What are the points to which you would direct your attention while accepting an appointment as an auditor of a company? State under what circumstances an appointed auditor can be removed from his...
-
State clearly the rights and duties of an auditor.
-
What are the special points to which an auditor should direct his attention for ascertaining the adequacy of provision for bad and doubtful debts in the context of proper valuation of sundry debtors?
-
What are the four financial statements that all public companies must produce?
-
What are the two components of a company's income tax provision? What does each component represent about a company's income tax provision?
-
Allene, CH2=C=CH2, has the structure shown below. Explain how the bonding in allene requires the two = CH2 groups at its ends to be at right angles to each other. allene
-
1. Draw the important resonance forms for each compound. 2. Label the hybridization and bond angles around each atom other than hydrogen. 3. Use a three-dimensional drawing to show where the...
-
List each set of compounds in order of increasing boiling point. (a) Hexane, octane, and decane (b) Octane, (CH3)3 C-C(CH3)3 and CH3CH2C(CH3)2CH2CH2CH3
-
Watch the recorded interview of Ms . Veronica Trammell, Executive Director of Learning Technology at KSU. The interview focused on service management and service operation. Share your thoughts in...
-
develop a detailed event plan following the assigned event type and theme with the following information: Event objectives based on the assigned event type and theme Two sets of varying food &...
-
Provide a detailed simulation performance results for Walmart inc. based on 5 year performance review.

Study smarter with the SolutionInn App



