Convert the following skeletal structures into molecular formulas, and tell how many hydrogens are bonded to each
Question:
Convert the following skeletal structures into molecular formulas, and tell how many hydrogens are bonded to each carbon: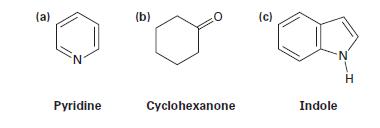
Transcribed Image Text:
(a) N Pyridine (b) Cyclohexanone (c) Indole N H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To convert the skeletal structures into molecular formulas and determine the number of hydrogens bon...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Tell the number of hydrogens bonded to each carbon atom in the following substances, and give the molecular formula ofeach: (c) (b) (a) Br. CEN
-
Tell the number of hydrogens bonded to each carbon atom in the following substances and give the molecular formula ofeach: OH H (a) (b) CO2CH3 Ephedrine Cocaine
-
How many hydrogens are replaced by deuterium when each of the following compounds is treated with NaOD in D2O? a. 3-methylcyclopentanone b. 3-methylhexanal
-
The figure below represents a schematic of pipe network. A rate of 35 Ls, is pumped to feed two lines (3-4-5-6; and 2-7-8). The length and diameter of each pipe segment are listed in the table....
-
Classify each of the following items as one of the following: Addition to the book balance (+ Book) Subtraction from the book balance ( Book) Addition to the bank balance (+ Bank) Subtraction from...
-
To initiate a nuclear reaction, an experimental nuclear physicist wants to shoot a proton into a 5.50-fm-diameter 12 C nucleus. The proton must impact the nucleus with a kinetic energy of 3.00 MeV....
-
1. To develop an understanding of your ethical leadership style 2. To understand how your preferred ethical leadership style relates to other ethical leadership styles Directions 1. Please read the...
-
Wallace Publishing identified the following overhead activities, their respective costs, and their cost drivers to produce the three types of textbooks the company publishes. Deluxe textbooks are...
-
The OLAP (Online Analytical Processing) discussion gives examples of multidimensional information. Give your own example of useful three-dimensional information.
-
Which of the butane conformations you drew Problem 2.13 do you think is the most stable? Explain. Problem 2.13 Looking along the C2-C3 bond of butane, there are two different staggered conformations...
-
Rank the substituents in each of the following sets: (a) CH3 -CHCCH3 CH3 (b) SH -NH -SO3H CH3 -CHCHCHCH3 -OCHCHOH -CH2CH2CH2CH2CH3
-
The CAE of PJS Company is working with senior management and the board to develop a combined assurance model and has asked you for advice. More specifically, he has asked you to respond to the...
-
Man SE is a German commercial vehicle manufacturer. Its DPS and EPS for 2011 were 2 and 4.62, respectively. The RoE of the firm was 11.85 per cent and the share price is 99.63. How would you...
-
How do differences in the macro-environment affect corporate governance?
-
Is it possible to improve one governance principle in a firm but weaken another at the same time? Use an illustration to explain your answer.
-
After extensive medical and marketing research, Pill plc believes it can penetrate the pain reliever market. It is considering two alternative products. The first is a medication for headache pain....
-
Why would we expect managers of a corporation to pursue the objectives of shareholders? What about bondholders?
-
Angela Petrillo recendy opened her own law office, which she operates as a corporation. The name of the new entity is Angela Petrillo, Attorney. Petrillo experienced the following events during the...
-
Do the three planes x + 2x + x 3 = 4, X X 3 = 1, and x + 3x = 0 have at least one common point of intersection? Explain.
-
Show the steps involved in the following reaction: CO,CH3 CO,CH3 + || + CO2 CO,CH3 CO,CH3
-
What is the chemical name of each of the following drugs? a. Benzocaine b. Procaine
-
What accounts for the ease of imine formation between penicillinase and the sulfone antibiotic that counteracts penicillin resistance?
-
Your Company enters into a finance lease as the lessee and determines the present value of the lease payments and residual value is $500,000. Your Company also incurs $8,000 in legal fees to execute...
-
Forced labor is immoral and should never be condoned. What other issues should companies be aware of that could exist in their supply chains? What are some examples of these issues and the companies...
-
Buildings $ 27,500 Accounts receivable 2,100 Land 16,200 Merchandise inventory 7,200 Accounts payable 5,100 Cash 8,200 Notes payable (due in 7 years) 32,000 Office supplies 1,200 Common stock 7,000...

Study smarter with the SolutionInn App


