Tell the number of hydrogens bonded to each carbon atom in the following substances and give the
Question:
Tell the number of hydrogens bonded to each carbon atom in the following substances and give the molecular formula ofeach:
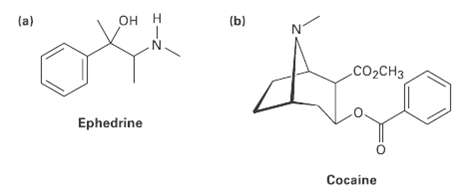
Transcribed Image Text:
OH H (a) (b) CO2CH3 Ephedrine Cocaine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (21 reviews)
2 2H 3H CH3 2 H 3H COCH...View the full answer

Answered By

Aun Ali
I am an Associate Member of Cost and Management Accountants of Pakistan with vast experience in the field of accounting and finance, including more than 17 years of teaching experience at university level. I have been teaching at both undergraduate and post graduate levels. My area of specialization is cost and management accounting but I have taught various subjects related to accounting and finance.
5.00+
13+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Tell how much hydrogen is bonded to each carbon in the following compounds, and give the molecular formula of each substance: (b) NHCH3 Adrenaline Estrone (a hormone)
-
Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (R) or (S) configuration. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH H CH,CH, H,C CH,CH, H H CH CCH, CI H...
-
Indicate the molecular geometry around each carbon atom in the compound H;CCH=CHCH,CCH,COOH
-
In what circumstances have courts opted to use the substantial-factor test rather than the but-for test?
-
John Benson, age 40, is single. His Social Security number is 111-11-1111, and he resides at 150 Highway 51, Tangipahoa, LA 70465. John has a 5-year-old child, Kendra, who lives with her mother,...
-
Explain how isoelectric focusing works.
-
A publisher finds that the mean number of grammatical errors per page of a book is six. Find the probability that the number of grammatical errors found on any given page is (a) exactly four, (b) at...
-
Blushing Brides, LLC, a publisher of wedding-planning magazines in Columbus, Ohio, opened an account with Gray Printing Co. in July 2000. On behalf of Blushing Brides, Louis Zacks, the firms...
-
How would you react to your instructor's suggestion to incorporate a data component into an assignment for your non-math-related course?
-
McCabe Oil Company has an account titled Oil and gas properties. McCabe paid $6,200,000 for oil reserves holding an estimated 500,000 barrels of oil. Assume the company paid $510,000 for additional...
-
The following molecular models are representations of (a) Adenine and (b) Cytosine constituents of DNA. Indicate the positions of the multiple bonds and lone pairs for both, and draw skeletal...
-
Identify the most electronegative element in each of the following molecules: (a) CH2FC1 (b) FCH2CH2CH2Br (c) HOCH2CH2NH2 (d) CH3OCH2Li
-
The Pew Research Center conducted a survey of randomly sampled American adults in 2008 and in 2010, asking them about their use of social networking sites such as Facebook. Table 7.31 shows age...
-
1. Who's Liable When a Recalled Product Causes an Injury? and why 2. what would happen if the product was never recalled and the person gets hurt?
-
What is operating leverage? How is operating leverage measured? What are the implications of operating leverage for the financial manager? What are the two parts of total return? Why are unrealized...
-
Imagine that you are a consultant for an investment fund which sold a client a call option on a stack. Explain to yen]: management {who are smart but not mathematically trained} what risk is present...
-
A listing of the MFH's ledger accounts as of March 31 is given below: Assets Cash Accounts receivable ($130,000 February sales; $1,600,000 March sales) Stock Prepaid insurance Property and equipment...
-
Strategy is developed across four different levels of an organization's hierarchy. What are those four levels and how do they differentiate from one another?
-
Calculate the net active return based on each possible gross active return provided using the selected data in Exhibit 1. Show your calculations. Jack Porter and Melissa Smith are co-managers for the...
-
A certain Christmas tree ornament is a silver sphere having a diameter of 8.50 cm. Determine an object location for which the size of the reflected image is three-fourths the size of the object. Use...
-
Nitrogen at 1 atm and 25C is to be compressed to 5 bar in an adiabatic, isentropic compressor. What is the temperature of the nitrogen stream exiting the compressor? If the flowrate of nitrogen is...
-
Explain which compound has the higher solubility in water.
-
Which of these compounds exhibit cis-trans isomerism? Draw both cis-trans isomers when they exist? a) CHCHCH=CHCH CH3 c) CHC=CHCH b) CH3CHCH=CH CI d) CHC=CHCHCH3
-
Draw the cis-trans isomers for these compounds and explain which is more stable: CH3 a) CH CH CHCOH NH c) e CH, CH2NHCH CH f CH3CH CH CH
-
Evaluate the challenges and solutions associated with implementing robust error handling and recovery mechanisms in operating systems, particularly in the context of disk I/O operations and system...
-
Erica, Deana & Tara had average capital balances of $350,000, $380,000 and $400,000 respectively during the current fiscal year. The partnership agreement provides for an allowance of 4% on the...
-
Retum to quest Grove Audio is considering the introduction of a new model of wireless speakers with the following price and cost characteristics. Sales price Variable costs Fixed costs Required: $...

Study smarter with the SolutionInn App


