Tell the number of hydrogens bonded to each carbon atom in the following substances, and give the
Question:
Tell the number of hydrogens bonded to each carbon atom in the following substances, and give the molecular formula ofeach:
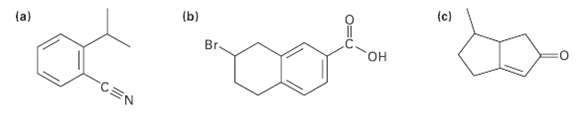
Transcribed Image Text:
(c) (b) (a) Br. он CEN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a 1 H 1 H 1 H 1H 1 OH 10...View the full answer

Answered By

Akhilandeswari Thiruneelakandan
I'm a mechanical engineer by profession and math enthusiast by passion. I love solving complex problems and I tutor in various online platforms like studypool and study.com. I am also an academic expert volunteer with an NGO Make A Difference, wherein I tutor 11th and 12th grade kids at a shelter home.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Tell how much hydrogen is bonded to each carbon in the following compounds, and give the molecular formula of each substance: (b) NHCH3 Adrenaline Estrone (a hormone)
-
Star (*) each asymmetric carbon atom in the following examples, and determine whether it has the (R) or (S) configuration. (a) (b) (c) (d) (e) (f) (g) (h) (i) CH H CH,CH, H,C CH,CH, H H CH CCH, CI H...
-
Indicate the molecular geometry around each carbon atom in the compound H;CCH=CHCH,CCH,COOH
-
Draw a graph showing the marginal revenue (MR) and marginal cost (MC) of increasing the tax rate (t) in the Peltzman model of regulation represented by equation (19.1). Place the tax rate on the...
-
Opal, Inc., is about to make its first attempt to borrow from a local bank. The company uses LIFO for tax purposes, solely to defer taxes, and believes that income computed using the FIFO method...
-
What is the sensitivity of the test for moderate-smoking students (students who smoke 1544 cigarettes per week)? Suppose the self-reports are completely accurate and are representative of the number...
-
Consider a short rate binomial lattice where the risk-free rate at \(t=0\) is \(10 \%\). At \(t=1\) the rate is either \(10 \%\) (for the upper node) or \(0 \%\) (for the lower node). Trace out the...
-
On January 1, 2010, Corgan Company acquired 80 percent of the outstanding voting stock of smashing, Inc., for a total of 980,000 in cash and other consideration, at the acquisition date, smashing had...
-
Pelican Paint Maufacturers, Inc. uses process costing for its mixing department. It mixed 5 , 0 0 0 units of paint during the period and has 6 0 0 are still being processed ( ending WIP ) in the...
-
The investor, before buying an apartment building, decided to estimate its cost. It is known that the areas of apartments are rented out, and at 90% of employment potential income per year is 170...
-
Convert the following structures into skeletal drawings: (a) (b) H. H. H. Indole 1,3-Pentadiene (d) (c) H. -CI C-CI H. 1,2-Dichlorocyclopentane Benzoquinone U
-
Propose structures for molecules that meet the following descriptions: (a) Contains two sp2-hybridized carbons and two sp3-hybrized carbons (b) Contains only four carbons, all of which are...
-
The Wagner Act, Taft-Hartley Act, and the Landrum-Griffin Acts provided the foundation for the rules and regulation of: A. Food processing B. Interstate food transport C. Labor relations D. Nutrition...
-
In light of these developments, what specific strategies or recommendations do you think auditors and audit firms should consider to ensure a seamless transition to these technological advancements...
-
In excel if you want to subtract the cost of fuel 150.00 in cell R6 from the selling price in cell W6 of 26 000.00 what should the calculation look lik?
-
Over next several weeks, your team will craft an article-quality executive summary about whether, why and how to modify a significant, proposed amendment to public company audit standards by the...
-
ACCT 6305 The concept of depreciation, amortization, and cost flow assumptions are new to the newly appointed CEO (who is a surgeon with decades of experience) of your organization. The CEO...
-
Janine is considering what auto costs she is going to have after buying a new car. She has budgeted enough money for the monthly auto loan payment, gas, and auto insurance. Has Janine factored in all...
-
Give an example of a law defining a crime that, although held to be constitutional by the U.S. Supreme Court, has been declared unconstitutional by some state courts. Why can such a result occur in...
-
Separate variables and use partial fractions to solve the initial value problems in Problems 18. Use either the exact solution or a computer-generated slope field to sketch the graphs of several...
-
A fluid obeys the equation of state a. For what values of the constants B and C will this fluid undergo a vapor-liquid phase transition? b. What is the molar internal energy change if this fluid is...
-
What is the hybridization at the N and each C in this molecule? Indicate the type of bond and the orbitals that are overlapping to form it for each of the designated bonds (for example, ?CSP3 + H1s)....
-
Consider hydrogen cyanide, H C N. (a) What is the hybridization at the N at the C? (b) What are the types of the three CN bonds? What orbitals overlapping to form them? (c) In what type of orbital...
-
What is the hybridization at each C in this molecule? Indicate the type of bond and the orbital's that are overlapping to form it for each of the designated bonds? ITT H=C=C=C=C_C7H (both) H tall...
-
Fixed-interest fund managers measure convexity in conjunction with duration to better measure their exposures to interest rate risk. Discuss the concept of convexity within the context of interest...
-
A friend owes you $500 in 1 year from now, and then $1000 2 years from now. Both loans are at 5%. If you want to replace both of these loans with ONE PAYMENT NOW, how much would your friend give you ?
-
If the amount of retained earnings at the beginning of the year was $ 3 0 , 0 0 0 , and $ 1 2 , 0 0 0 in dividends is paid during the year, calculate net income for the year.

Study smarter with the SolutionInn App


