Draw the enantiomer, if any, for each structure. a. b. c. d. e. f. g. h. CH3
Question:
Draw the enantiomer, if any, for each structure.
a.
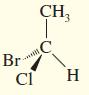
b.
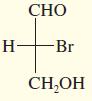
c.
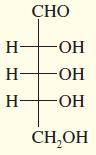
d.
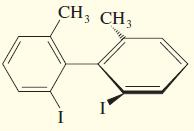
e.
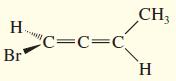
f.
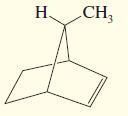
g.
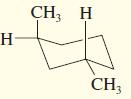
h.
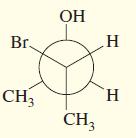
Transcribed Image Text:
CH3 Br H C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
a b c d e f Do...View the full answer

Answered By

Vartika ..
Having secured 10 CGPA during school level education, and being one of the toppers in my school as well as being college topper with distinction, I have always conceptualised chemistry not just as a subject, but also as a mystery to explore more about.
I have been teaching since college days and have helped students clear their doubts as well as exams.
The biggest compliment i received was when one of my students said that I answer even those queries, which even their teachers couldn't answer.
I welcome you all to come with me and explore the mysterious world of chemistry ;)
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the enantiomer for each of the following compounds: a. b. c. d. e. f. g. h. i. j. k. l . CI.
-
Draw both conformations for each of the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) Br
-
Predict the products, if any, of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (1) LiAIH (2) HO COOH (1) NaCN (2) H,0, heat CH,Br (1) SoCh 2) AIC COOH KMnO,. Hho (warm, concd.)...
-
Determine one positive and one negative coterminal angle for each angle given. 173
-
Show that the limiting fraction of association of a base in water, as the concentration of base approaches 0, is = 107 Kb/ (1 + 107Kb). Find the limiting value of for Kb = 10-4 and for Kb = 10-10.
-
Nitrogen at 100 psia and 300F in a rigid container is cooled until its pressure is 50 psia. Determine the work done and the heat transferred during this process, in Btu/lbm.
-
Your uncle has given you a bond that will pay $\$ 500$ at the end of each year forever into the future. If the market yield on this bond is $8.25 \%$, how much is it worth today?
-
Air travel on Mountain Airlines for the past 18 weeks was: a. Explain why an averaging technique would not be appropriate for forecasting. b. Use an appropriate technique to develop a forecast for...
-
Pharoah Company uses a job order cost system in each of its three manufacturing departments. Manufacturing overhead is applied to jobs on the basis of direct labor cost in Department D, direct labor...
-
Kaleidoscope Cutlery manufactures kitchen knives. One of the employees, whose job is to cut out wooden knife handles worked 49 hours during a week in January. The employee earns $14 per hour for a...
-
Give the stereochemical relationships between each pair of structures. Examples are same compound, structural isomers, enantiomers, diastereomers. Which pairs could you (theoretically) separate by...
-
Label each structure as Z, E, or neither. a. b. c. d. H,C CH, C=C H CI
-
Using the following data from the comparative balance sheet of Doabe Company, illustrate horizontalanalysis. December 31, 2013 $ 520,000 $ 840,000 December 31, 2012 $ 400,000 $ 600,000 Accounts...
-
You have been asked to create a team of four people within an organization to develop an internal company blog. Your role is to create an environment that supports the proactive use of the blog by...
-
The managing director (MD) of a medium-sized company experiencing difficulties at the beginning of an economic downturn wrote the following message on the blog used by employees. Using the principles...
-
How have market globalization and cultural diversity contributed to the increased importance of intercultural communication?
-
Look through recent issues (print or online) of Bloomberg Businessweek, Fortune, or other business publications for an article that describes how an executives conclusions about his or her companys...
-
The Securities and Exchange Commission (SEC) requires every public company to file a comprehensive financial report known as Form 10-K every year. Companies usually combine this highly structured...
-
Why might a corporation purchase debt or equity securities of another company?
-
In Exercises 15 through 30, find the derivative dy/dx. In some of these problems, you may need to use implicit differentiation or logarithmic differentiation. y ex + et -2x 1 + e
-
Give a single structure that fits the data provided. A compound C5H9C13 has three NMR absorptions at 1.99, 4.31, and 8 6.55 with relative integrals of 6:2:1, respectively.
-
Give a single structure that fits the data provided. A compound C5H9C13 has three NMR absorptions at 1.99, 4.31, and 8 6.55 with relative integrals of 6:2:1, respectively.
-
Strange results in the undergraduate organic laboratory have led to the admission by a teaching assistant, Thumbs Throckmorton, that he has accidentally mixed some terf-butyl bromide with the methyl...
-
The purpose of this assignment is to better understand what the important pieces of the Linux/Unix operating system that you might want to prioritize reviewing when you are doing a forensic...
-
What is the function of the Helix system? What are the benefits of this tool? How does it work in terms of digital evidence collection? How do you get evidence of Unix, Linux, and Mac OS operating...
-
Analyzing revenue, expense, and withdrawal transactions into debit and credit parts Use the chart of accounts for Hals Marketing Services given in Application Problem 2-2. Transactions: Mar. 25. Sold...

Study smarter with the SolutionInn App


