How many absorptions would you expect the following compound to have in its 13C NMR spectrum?
Question:
How many absorptions would you expect the following compound to have in its 13C NMR spectrum?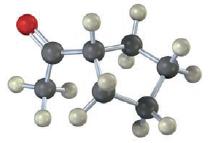
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
Based on the provided image the compound appears to be 2bromopropane also known as isopr...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many absorptions would you expect the following compound to have in its 1H and 13C NMRspectra?
-
Suppose you ran a DFPT-135 spectrum for each substance in Problem 13.33. Which carbon atoms in each molecule would show positive peaks and which would show negative peaks? Problem 13.33 How many...
-
How many 13C NMR absorptions would you expect for cis-1, 3-dimethyl- cyclohexane for trails-1, 3-dirnethylcyclohexane? Explain.
-
The table shows equivalent temperatures in degrees Celsius and degrees Fahrenheit. (a) Plot the data with Fahrenheit temperature on the x-axis and Celsius temperature on the y-axis. What type of...
-
Megatronics Corporation, a massive retailer of electronic products, is organized in four separate divisions. The four divisional managers are evaluated at year-end, and bonuses are awarded based on...
-
Solid Solutions Inc. is considering the purchase of automated machinery that is expected to have a useful life of five years and no residual value. The average rate of return on the average...
-
Consider the IRR and ERR measures of worth. If we define a root to mean a value for the measure that results in \(\mathrm{PW}=0\), then which of the following statements is true? a. Both IRR and ERR...
-
Healthy Products, Inc., uses a traditional product costing system to assign overhead costs uniformly to all products. To meet Food and Drug Administration requirements and to assure its customers of...
-
Country A is endowed with 18 units of good x and 1 unit of good y, while country B is endowed with 2 units of good x and 7 units of good y. Both countries consume x and y in fixed and equal...
-
Propanil, marketed under names such as Stampede and Chem-Rice, is commonly used to prevent weeds in rice fields. How would you prepare propanil from 3,4-dichloroaniline? Show the mechanism of your...
-
How many peaks would you expect in the 13C NMR spectra of the following compounds? (a) (d) (b) HC. CH3 (e) CH3 (c) (f) H3C HC CH3 CH3 CHCH3 CH3
-
In 1970, Amos Flora of Flora, Indiana, discovered teeth and jawbones while dredging a creek. Scientists discovered that the bones were from a mastodon and that they had lost 77.2% of their carbon-14....
-
Federated Tools, Inc., charges Jacks Hardware five cents per item and Eves Home Store ten cents per item for the same product. Jacks Hardware and Eves Home Store are competitors. If this practice...
-
Auto Corporation makes cars in the United States. To boost the sales of Auto Corporation and other domestic carmakers, Congress can a. neither set quotas nor tax imports. b. only set quotas on...
-
Digital, Inc., makes supercomputers that feature advanced technology. To inhibit Digitals export of its products to other countries, Congress can a. confiscate all profits on exported supercomputers....
-
Under a force majeure clause, a party may be excused from liability for nonperformance. (True/False)
-
Kenya issues bonds to finance the construction of an international airport. Kenya sells some of the bonds in the United States to Larry. A terrorist group destroys the airport, and Kenya refuses to...
-
Write the accounting equation.
-
2.) Find the Laplace transform of f(t) 7e-St cos 2t +9 sinh2 2t. Use Laplace Table. %3D
-
Compare the Lewis dot structure of nitrogen and phosphorus and explain why you might expect these two atoms to exhibit similar bonding properties?
-
Predict the products that are expected when each of the following alkenes is treated with ozone followed by DMS: a. b. c. d. e. f.
-
Draw the products obtained when 3,3,6-trimethylcyclohexene is treated with NBS and irradiated with UV light.
-
Apply the Ke formula that you learned in the chapter to a stock from a US corporation that is currently trading. Use a constant growth rate of 4%. Explain what you have discovered
-
Liability insurance is First-party insurance. Fourth-party insurance. Third-party insurance. Second-party insurance. best answer?
-
Kerstin Dugmore plans to send their kids to college in 4 years and will need another $13,420.00 before they can do so. Given an interest rate of 1.00% compounded tri-annually, how much money do they...

Study smarter with the SolutionInn App


