How many peaks would you expect in the 13C NMR spectra of the following compounds? (a) (d)
Question:
How many peaks would you expect in the 13C NMR spectra of the following compounds?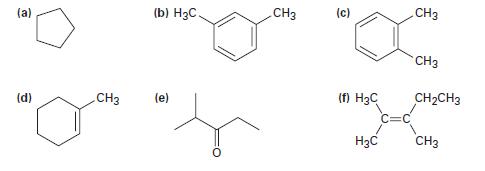
Transcribed Image Text:
(a) (d) (b) H₂C. CH3 (e) CH3 (c) (f) H3C H₂C CH3 CH3 CH₂CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To determine the number of peaks in the 13C NMR spectra of the given compounds we need to consider t...View the full answer

Answered By

Abdul Wahab Qaiser
Before working at Mariakani, I volunteered at a local community center, where I tutored students from diverse backgrounds. I helped them improve their academic performance and develop self-esteem and confidence. I used creative teaching methods, such as role-playing and group discussions, to make the learning experience more engaging and enjoyable.
In addition, I have conducted workshops and training sessions for educators and mental health professionals on various topics related to counseling and psychology. I have presented research papers at conferences and published articles in academic journals.
Overall, I am passionate about sharing my knowledge and helping others achieve their goals. I believe that tutoring is an excellent way to make a positive impact on people's lives, and I am committed to providing high-quality, personalized instruction to my students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many signals would you expect in the 13 C NMR spectrum of each of the compounds in Problem 16.34? In Problem 16.34 How many signals would you expect in the 1 H NMR spectrum of each of the...
-
How many signals do you expect in the 13 C NMR spectrum of each of the following compounds? a. b. c. d. Br
-
Suppose you ran a DFPT-135 spectrum for each substance in Problem 13.33. Which carbon atoms in each molecule would show positive peaks and which would show negative peaks? Problem 13.33 How many...
-
Solve the equation (a) Graphically, (b) Numerically, and (c) Symbolically. Then solve the related inequality. |4x7| = 5, |4x - 7| 5
-
Refer to the data for problem 1336 regarding Omaha Grain Company. In problem 1336, Omaha Grain Company has two divisions, which reported the following results for the most recent year. Required:...
-
The total factory overhead for Maritime Marine Company is budgeted for the year at $600,000. Maritime Marine manufactures two types of boats: speedboats and bass boats. The speedboat and bass boat...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Mammoth Corporation is considering a 3-for-2 stock split. It currently has the stockholders equity position shown. The current stock price is $120 per share. The most recent periods earnings...
-
Risk of entry Bargaining power of suppliers Bargaining power of buyers Threat of substitutes Power of complement providers Rivalry among established firms in industry Which of the above forces are...
-
How many absorptions would you expect the following compound to have in its 13C NMR spectrum?
-
In light of your answer to Problem 12.47, which would you expect to be more basic, aniline or p-methoxyaniline? Explain. Problem 12.47 How can you explain the observation that p-nitroaniline is less...
-
The table shows the heights (in inches) and weights (in pounds) of 14 college men. The scatterplot shows that the association is linear enough to proceed. a. Find the equation for the regression line...
-
The foundation of international space law is the Convention on International Liability for Damage Caused by Space Objects. (True/False)
-
A contract between Moss Energy, a U.S. firm, and Electronique, S.A., a French company, provides that disputes between the parties will be adjudicated in a specific British court. This clause is a. a...
-
A survey indicated that chocolate is the most popular flavor of ice cream in America. For each of the following, indicate the possible effects on demand, supply, or both as well as equilibrium price...
-
A South African seller and a U.S. buyer form a contract, which the buyer later breaches. The seller sues in a South African court and wins damages, but the buyers assets are in the United States. If...
-
Johnston International, a U.S. firm, signs a contract with Irkut, Ltd., a Russian company, to give Irkut the right to sell Johnstons products in Russia. This is a. a distribution agreement. b. a...
-
What are the three basic financial statements illustrated in the text.
-
Fahrad Inc. sells all of its product on account. Fahrad has the following accounts receivable payment experience: Percent paid in the month of sale .........10 Percent paid in the month after the...
-
Draw all possible stereo-isomers for each of the following compounds. Each possible Stereo-isomer should be drawn only once: a. b. c. d. e.
-
Identify the structure of the starting alkene in each of the following cases: a. b. c. 1) O3 C3H14 2) DMS C10H16 1) 03 2) DMS
-
When 2-methylpropane is treated with bromine in the presence of UV light, one product predominates. (a) Identify the structure of the product. (b) Draw the structure of the expected minor product....
-
Property insurance is considered Second-party insurance Third party insurance First party insurance Fourth party insurance best answer?
-
Arppel Company uses a periodic inventory system and closes its accounts annually on 31 December. Below are extracted from the books of the Company: Account balance as at 31 Dec 2020 Inventory $42,000...
-
Bill Board must choose between two bonds: Bond A pays $75 annual interest with semiannual payment and has a market value of $840. It has 8 years to maturity. Bond B pays $65 annual interest with...

Study smarter with the SolutionInn App


