Imidazole, which forms part of the structure of the amino acid histidine, can act as both an
Question:
Imidazole, which forms part of the structure of the amino acid histidine, can act as both an acid and a base. Look at the electrostatic potential map of imidazole, and identify the most acidic hydrogen atom and the most basic nitrogen atom.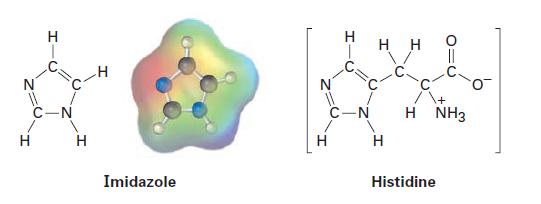
Transcribed Image Text:
I C-N H H Imidazole N 11 C-N HH H H NH3 Histidine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Imidazole is a fivemembered ring compound with three carbon atoms and two nitrogen atoms One ...View the full answer

Answered By

FELIX NYAMBWOGI
I have been tutoring for over 5 years, both in person and online. I have experience tutoring a wide range of subjects, including math, science, English, and history. I have also worked with students of all ages, from elementary school to high school.
In addition, I have received training in effective tutoring strategies and techniques, such as active listening, questioning, and feedback. I am also proficient in using online tutoring platforms, such as Zoom and Google Classroom, to effectively deliver virtual lessons.
Overall, my hands-on experience and proficiency as a tutor has allowed me to effectively support and guide students in achieving their academic goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Imidazole forms part of the structure of the amino acid histidine and can acts as both an acid and a base (a) Look at the electrostatic potential map of Imidazole, and identify the most acidic...
-
Lawby Joseph Lowry and why Shariah? by Noah Feldman illustrate the constitutions, judicial institutions and certain legal cod Shariah Law in Arb world. Arab Legal System Arab legal system forms part...
-
Mouse liver cells were homogenized and the homogenate subjected to equilibrium density-gradient centrifugation with sucrose gradients. Fractions obtained from these gradients were assayed for marker...
-
Three resistors are connected in series across a battery. The value of each resistance and its maximum power rating are as follows: 2.0 and 4.0 W, 12.0 and 10.0 W, and 3.0 and 5.0 W. (a) What is...
-
The following information is for funds used to account for the transactions of the Hospital of Havencrest, which is operated by a religious organization. The balances in the accounts are as of June...
-
What are the main factors that have created positive network externalities for eBay?
-
Many single women and married couples use donated sperm to conceive children each year. Pennsylvania resident Donna Donovan decided to use donated sperm from Idant Laboratories, a New York sperm bank...
-
Drilling Innovations, Inc., produces specialized cutting heads used by companies that drill for oil and natural gas. The company has recently implemented an ABC system for three of its products and...
-
Q1) Determine whether the roots of the each quadratic are real or nonreal and whether they are equal or unequal. (5 marks) a) 2x+3x-2=0 b) 4x+4x+1=0 Q2) Solve for X by factoring: 3x(x-3) + 22 = 2x +...
-
Draw a tetrahedral representation of tetrachloromethane, CCl 4 , using the standard convention of solid, dashed, and wedged lines.
-
Show how the species in part (a) can act as Lewis bases in their reactions with HCl, and show how the species in part (b) can act as Lewis acids in their reaction with OH - . (a) CH3CHOH, (CH3)2NH,...
-
One important character in rosids is the presence of named for the rose family, Rosaceae, and this one family has many genera that produce fruits that we like to eat. Name the genera of the following...
-
If a projectile with a mass of 9.30 g is traveling at a speed of 1.26 km/s, determine the following. (a) the kinetic energy (in kJ) of the projectile in kilojoules kJ (b) the kinetic energy (in kJ)...
-
Analyze how social media marketing can empower a marketing strategy, including the benefits and potential risks (if any) of using this type of marketing. Explain how search and content marketing can...
-
CASE STUDY: A noteworthy and historic case of involuntary active euthanasia, with ramifications that persist today, is that of Robert Latimer, a Saskatchewan farmer who killed his daughter, Tracy, in...
-
A Del Monte bond with a face value of $1,000 has an annual coupon of 6.75%. The bond had an original 7-year tenor and 4 years remaining to maturity. If the market interest rate for similar high yield...
-
Please write about social media and the bullying, harassment, and discrimination that takes place. Use a real world lawsuit you find about someone suing someone for online harassment and talk about...
-
A multiple-choice test has nine questions. For each question there are four possible answers from which to select. One point is awarded for each correct answer, and points are not subtracted for...
-
Periwinkle Company is a multinational organization. Its Parts Division is located in Lavender Land, while its Assembly Division is located in North Orchid. During the current year Periwinkle Companys...
-
Upon treatment with ozone followed by work-up with hydrogen peroxide, an alkene forms formic acid and a compound that shows three signals (a singlet, a triplet, and a quartet) in its 1 H NMR...
-
A compound forms cis-1,2-dimethycyclopropane when it is reduced with H 2 and Pd/C. The 1 H NMR spectrum of the compound shows only two singlets. What is the structure of the compound?
-
Compare the mechanisms of the following reactions: NH2 + NH2 + CI `N CI NH, NH, + CF
-
N A long current-carrying wire, oriented North-South, lies on a table (it is connected to batteries which are not shown). A compass lies on top of the wire, with the compass needle about 3 mm above...
-
2. The accompanying figure shows a current loop consisting of two concentric circular arcs and two perpendicular radial lines. Determine the magnetic field at point P. b a-
-
Part A The 8-kg sack slides down the smooth ramp. It has a speed of 1.5 m/s when y = 0.32 m. Suppose that y(x)=0.32e m, where x is in m. (Figure 1) Determine the magnitude of the normal reaction the...

Study smarter with the SolutionInn App


