List the following species in order of decreasing nucleophilicity in an aqueous solution: CH;OH HO CH;CO CH3S
Question:
List the following species in order of decreasing nucleophilicity in an aqueous solution:
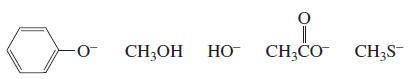
Transcribed Image Text:
CH;OH HO CH;CO CH3S
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
Nucleophilicity Nucleophilicity increases as the charge on the atom increases In aqueous solution fo...View the full answer

Answered By

Akshay Nagar
Experience:-
I have 2 years of experience of teaching Physics to class understudies as well as other plateforms.In my day to day classes I teach students about Numericals,Concepts,Data Collection,Exam preparation and a whole spectrum of Physical Science.
Qualifications:-
1. Bachelor of Science with Maths and Physics major (MDU,Rohtak, India)
2. Bachelor of Education (CBLU, Bhiwani,India)
3. Masters of physics (MDU, Rohtak, India)
4. Teach English certified teacher (Arizona state university,Tempe, Arizona)
5. Specialization in Electronic Music Production (Berklee College Of Music, Boston,Massachusetts)
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
List the following species in order of decreasing basicity: a. b. CH,CHC CHCH NH2 CH,CH20 F
-
List the following compounds in order of decreasing reactivity in an E2 reaction: CH CD CH CH3 CD
-
List the following compounds in order of decreasing boiling point: OH NH2 HO
-
A PLC is used to count the number of cans traveling by on a conveyor belt in a fish canning factory. An optical proximity switch detects the passage of each can, sending a discrete (on/off) signal to...
-
A measurement with a signal-to-noise ratio of 100/1 can be thought of as a signal, S, with 1% uncertainty, e. That is, the measurement is S e = 100 1. (a) Use the rules for propagation of...
-
Dahlia Simmons, CFO of Ulrich Enterprises, is analyzing a new project to sell solar-powered batteries for cell phones. Dahlia has estimated the following probability distributions for the variables...
-
Split the Bill? When the time comes for a group of people eating together at a restaurant to pay their bill, sometimes they might agree to split the costs equally and other times will pay...
-
Why do some transactions require an entry in a fund and another in an account group? What impact would there be if a journal entry were made only in the fund or only in the account group?
-
The salesman at the lot of Smiles Pre-Owned Vehicles told the woman buying the car that it was in "mint condition." Is this an express warranty? Why or why not?
-
Charles Price, a connoisseur of fine chocolate, opened Charles Fine Chocolate Co. in Stowe, VT on February 1, 2019, by investing $30,000 in cash into the business. The shop specializes in a selection...
-
a. Which is a stronger base, RO - or RS - ? b. Which is a better nucleophile in an aqueous solution?
-
What product is obtained when ethylamine reacts with excess methyl iodide in a basic solution of potassium carbonate? K,CO3 CH;CH,NH, + CH3-I excess
-
An article in the New York Times states, "On Aug. 15, 1971, President Nixon unhitched the value of the dollar from the gold standard." Is the author of this article correct that the United States...
-
Why a case study on Amazon? Surely everyone knows about Amazon and what it does? Yes, well, thats maybe true, but this case goes under the surface to review some of the insider secrets of Amazons...
-
Review the options for targeting particular audience groups online with different digital media.
-
The 4 Ps are all a marketing manager needs to create a marketing strategy for a service business. Prepare an argument that opposes this statement, and support it with examples.
-
Describe the difference between high-contact and low-contact services, and explain how the nature of a customers experience may differ between the two.
-
How can an organisation vary its promotional mix using the Internet?
-
Etobicoke Enterprises is deciding whether to expand its production facilities. Although long-term cash flows are difficult to estimate, management has projected the following cash flows for the first...
-
Could a set of three vectors in span all of? Explain. What about n vectors in when n is less than m? R4
-
(a) In most peptides, the amide bonds have the Z conformation; explain why. (b) One particular amino acid residue in the PepC position adopts the E conformation in some cases. Which amino acid...
-
When N - acetyl- L -aspartic acid is treated with acetic anhydride, an optically active compound A, C6H7NO4, is formed" Tleaffient of A with the amino acid L -alanine yields two separable, isomeric...
-
When N - acetyl- L -aspartic acid is treated with acetic anhydride, an optically active compound A, C6H7NO4, is formed" Tleaffient of A with the amino acid L -alanine yields two separable, isomeric...
-
Discuss the dynamic organization of the cytoskeleton and its pivotal role in cellular motility, intracellular transport, and structural integrity .
-
Would you support the idea of a government issued Digital currency? Why ? and why not?
-
To protect her savings against further inflation and to help her prepare for a healthy financial future, Hanna Lind deposits $9,100 in an investment account earning 6% interest compounded quarterly....

Study smarter with the SolutionInn App


