What product is obtained when ethylamine reacts with excess methyl iodide in a basic solution of potassium
Question:
What product is obtained when ethylamine reacts with excess methyl iodide in a basic solution of potassium carbonate?
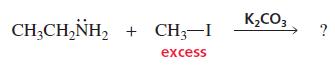
Transcribed Image Text:
K,CO3 CH;CH,NH, + CH3-I excess
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)

Answered By

Shubham Belsare
I'm currently pursuing Post graduation in organic chemistry and I'm passionately driven to teach Organic Chemistry to the students who finds hard time learning the subject. Apart from my regular college, I'm also a part-time subject expert on Chegg and I very much like and enjoy clearing the doubts of students which also increase my knowledge toward the subject.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the major product(s) that are expected when each of the following amines is treated with excess methyl iodide and then heated in the presence of aqueous silver oxide. (a) (b) NH2 NH2
-
Only a substitution product is obtained when the following compound is treated with sodium methoxide: Explain why an elimination product is not obtained. CH3 Br CH3
-
What products would be obtained when acetophenone reacts under each of the following conditions? (a) (b) (c) (d) (e) (f) (g) Acetophenone HNO H2S04 CBH NHNH2, HA NaBH CH2OH (1) CBHMgBr (2) NHC...
-
(2) Use Figure 10.2 shows a multi-degree-of-freedom (MDOF) model of three connected masses. Find the following for the MDOF system in Figure 10.2: Find expressions for the kinetic energy (T) and...
-
Explain how the difference voltage in Figure 19-35 reduces noise from source flicker.
-
You have been at your job with East Coast Yachts for a week now and have decided you need to sign up for the companys 401(k) plan. Even after your discussion with Sarah Brown, the Bledsoe Financial...
-
Diet Cola and Weight Gain in Humans A study found that American senior citizens who report drinking diet soda regularly experience a greater increase in weight and waist circumference than those who...
-
Phil Emley owns a garage and is contemplating purchasing a tire retreading machine for $18,000. After estimating costs and revenues, Phil projects a net cash flow from the retreading machine of...
-
XYZ Limited is a listed company on the Jamaica Stock Exchange. Its market capitalization as at December 3 1 , 2 0 1 5 was $ 5 5 0 m and the outstanding shares at the same date was 4 0 , 0 0 0 , 0 0 0...
-
1. What is the appropriate Box-Jenkins model to use on the original data? 2. What are your forecasts for the first four weeks of January 1983? 3. How do these forecasts compare with actual sales? 4....
-
List the following species in order of decreasing nucleophilicity in an aqueous solution: CH;OH HO CH;CO CH3S
-
Most of the pK a values given throughout this text are values determined in water. How would the pK a values of the following classes of compounds change if they were deter-mined in a solvent less...
-
Create an original example to demonstrate how an individual or firm acting out of self-interest (seeking profits by offering goods or services in economic markets) must benefit consumers even if...
-
What are the five powerful forces transforming the service landscape, and what impact do they have on the service economy?
-
Discuss the extent to which B2B e-marketplaces are fundamentally different from traditional offline markets.
-
Which factors are important in selecting a web design agency?
-
Give three reasons explaining why a website may have to integrate with existing marketing information systems and databases within a company.
-
Which techniques can be used to promote a website in offline media?
-
Explain why picking the project with the highest NPV might not be optimal when you evaluate projects with different resource requirements?
-
Find a least expensive route, in monthly lease charges, between the pairs of computer centers in Exercise 11 using the lease charges given in Figure 2. a) Boston and Los Angeles b) New York and San...
-
Lysozyme is an antibacterial enzyme that hydrolyzes polysaccharides in bacterial cell walls. It also catalyzes the hydrolysis of a -1,4-linked hexasaccharide oligomer of N-acefylglucosamine into a...
-
Lysozyme is an antibacterial enzyme that hydrolyzes polysaccharides in bacterial cell walls. It also catalyzes the hydrolysis of a -1,4-linked hexasaccharide oligomer of N-acefylglucosamine into a...
-
Classify the following peptides as acidic, basic, or neutral. What is the net charge on each peptide at pH = 6? (a) Gly-Leu-Val (b) Leu-Trp-Lys-Gly-Lys (c) N-acetyl-Asp-Val-Ser-Arg-Arg (A-acetyl...
-
What theorists are most likely to argue the city's form and growth come from decisions made by people and organizations that control wealth and other key resources?
-
Explain the difference between glucogenic and ketogenic amino acids
-
Describe what is an arrangement where workers who don't join a union must make payments equal to union dues and fees to get union representation services?

Study smarter with the SolutionInn App


