Show the products of each of the following reactions: (a) (b) (c) || CH3CHCHCHCHCSCOA Product of (a)
Question:
Show the products of each of the following reactions: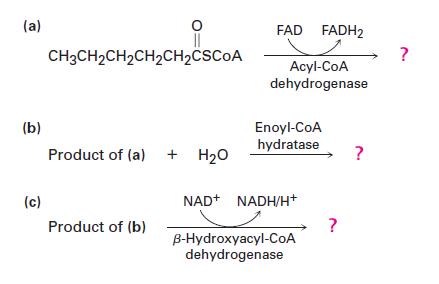
Transcribed Image Text:
(a) (b) (c) || CH3CH₂CH₂CH₂CH₂CSCOA Product of (a) + H₂O Product of (b) FAD FADH2 Acyl-CoA dehydrogenase Enoyl-CoA hydratase NAD+ NADH/H+* B-Hydroxyacyl-CoA dehydrogenase ? ? ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a AcylCoA dehydrogenase The product is enoylCoA The FAD cofactor is reduced to FADH2 b EnoylCoA hydr...View the full answer

Answered By

Rahul Rajpoot
"I am a BSC (Bachelor of Science ) from SGBAU India and also have a medical certificates from The Sri Guru Medical Panjab University, Panjab
LEARNING IS PASSION ACHIVESOMTING GREATFULL IDEAS
MOST OF THE I AM A RESERACHER OF BARC
I AM ALSO MBBS FIRST YAER PASS OUT MEDICAL STUDENT
NOW I AM PURSUING 2ND YEAR.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the products of each of the following reactions indicated by the curved-arrow notation. (a) (b) CH CH NH2
-
Show the products of the reactions of these carboxylic acids with PB3/Br2 before and after hydrolysis. (a) Pentanoic acid (b) Phenylacetic acid (c) Succinic acid (d) Oxalic acid
-
Give the products of each of the following reactions (ignore stereoisomers): a. b. CH3
-
The costs of achieving emission reductions in the future will depend greatly on the types of policies used to reduce emissions today. Explain.
-
Rocky Mountain Tire Center sells 20,000 go-cart tires per year. The ordering cost for each order is $40, and the holding cost is 20% of the purchase price of the tires per year. The purchase price is...
-
What is corporate governance? What are the areas of corporate governance discussed in the Qantas Group annual report?
-
Give advantages of two-stroke engine over four stroke engine.
-
Comment on the validity and reliability of the following: a. A respondent's report of an intention to subscribe to Consumer Reports is highly reliable. A researcher believes that this constitutes a...
-
Magic Mountain retires its 9% bonds for $128,000 before their scheduled maturity. At the time, the bonds have a face value of 126,000 and a carrying value of $117,000. Record the early retirement of...
-
Let ni, n 2 , ... , n 2 , n t be positive integers such that n 1 + n 2 + + n t = n and for each i let M i be an n i X n i matrix. Let M be then X n matrix where the main diagonal of each M i lies...
-
Predict the splitting patterns for each proton in the following molecules: (a) CHBr2CH3 (d) || CH3CHCOCHCH3 CH3 (b) CH3OCHCHBr (e) CH3CHCOCHCH3 CH3 (c) CICHCHCHCI (f)
-
Steam flows through a pipe of diameter 5 cm with a velocity of 50 m/s at 500 kPa. If the mass flow rate of steam is measured at 0.2 kg/s, determine (a) The specific volume (v) of steam in m3/kg. (b)...
-
Let \(B\) be a BM and \(M_{t}^{B}:=\sup _{s \leq t} B_{s}\). Let \(f(t, x, y)\) be a \(C^{1,2,1}\left(\mathbb{R}^{+} \times \mathbb{R} \times \mathbb{R}^{+} ight)\)function such that...
-
Prove that \(\left(m_{s}, s \geq 0 ight)\) is unique in \(L_{l o c}^{2}(X)\).
-
If two martingales are independent, they are orthogonal. Check that the converse does not hold. Let \(B\) and \(W\) be two independent Brownian motions. The martingales \(W\) and \(M\) where...
-
Let \(W=\left(W^{1}, \ldots, W^{d} ight)\) be a \(d\)-dimensional BM. Is the space of martingales \(\sum_{i=1}^{d} \int_{0}^{t} H_{i}\left(W^{i}{ }_{.} ight)_{s} d W_{s}^{i}\) dense in the space of...
-
Prove that, for \(\lambda>0\), one has \[\int_{0}^{\infty} e^{-\lambda t} p_{t}(x, y) d t=\frac{1}{\sqrt{2 \lambda}} e^{-|x-y| \sqrt{2 \lambda}}\] Prove that if \(f\) is a bounded Borel function, and...
-
A cylinder with an initial volume of 10.0 L is fitted with a frictionless piston and is filled with 1.00 mole of an ideal gas at 25oC. Assume that the surroundings are large enough so that if heat is...
-
The rate at which the temperature of an object changes is proportional to the difference between its own temperature and the temperature of the surrounding medium. Express this rate as a function of...
-
Identify the reagents that you would use to achieve each of the following transformations: a. b. Br Br
-
Discuss the following statement: Heating an object causes its temperature to increase.
-
The Joule coefficient is defined by (T /V) U = (1/C V )[P T(P/T) V ]. Calculate the Joule coefficient for an ideal gas and for a van der Waals gas.
-
Find all relative extrema of the function. (If an answer does not exist, enter DNE.) f(x)=-5x2+7x+3 relative maximum (x, y) = X relative minimum (x, y) = (dne
-
On the axis below, sketch the graph of a function that satisfies all of the following conditions: Vertical Asymptote at x=0 f'(x) > 0 if
-
Find the absolute extrema of the function on the closed interval. Use a graphing utility to verify your results. (If an answer does not exist, enter DNE.) f(x) = x+4x-1, [-2, 2] absolute maximum (x,...

Study smarter with the SolutionInn App


