Show the products of hydrolysis of the following esters: (a) 0 CH3 || | CH3COCHCH3 (b) COCH3
Question:
Show the products of hydrolysis of the following esters: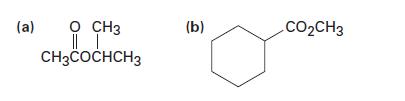
Transcribed Image Text:
(a) 0 CH3 || | CH3COCHCH3 (b) CO₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
To determine the products of hydrolysis for the given esters we need to break down the ester bonds u...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The hydrolysis of sulfonate esters of 2-octanol is a stereospecific reaction and proceeds with complete inversion of configuration. Write a structural formula that shows the stereochemistry of the...
-
The hydrolysis of sulfonate esters of 2-octanol is a stereospecific reaction and proceeds with complete inversion of configuration. Write a structural formula that shows the stereochemistry of the...
-
Show what esters would undergo Claisen condensation to give the following b-keto esters. (a) (b) (c) CH,CHCH,-C" - CH,CH,-CH-C-OCH,CH, Ph-CH,-C" O Ph CHCOCH CH(CH32
-
In Problems 1130, solve each equation by factoring. x 2 - 9x = 0
-
Assume that Department A has a higher contribution margin ratio but a lower responsibility mar gin ratio than Department B. If $10,000 in advertising is expected to increase the sales of either...
-
What steps should be taken by an organization that is committed to shattering the glass ceiling?
-
FRAUD PREVENTION AND DETERRENCE IN ACTION Assume the following facts: thirty-day collection period. The invoice is used to post-sales to the accounting system. Checks are received in the mailroom and...
-
The cash account for Ambulance Systems at February 29, 2012, indicated a balance of $20,580. The bank statement indicated a balance of $24,750 on February 29, 2012. Comparing the bank statement and...
-
4. A monopolist is faced with the inverse demand function P(Q) denoting the price when output is Q. The monopolist has a constant average cost k per unit produced. (a) Find the profit function (Q),...
-
How would you prepare the following ketones by reaction of a Grignard reagent and a nitrile? (a) || CH3CH2CCH2CH3 (b) ON CH3
-
The reduction of an amide with LiAlH 4 to yield an amine occurs with both acyclic and cyclic amides (lactams). What product would you obtain from reduction of 5,5-dimethylpyrrolidin-2-one with...
-
Why are throttling devices commonly used in refrigeration and air-conditioning applications?
-
In January 2019, Leon McLeod received a gift of a beach cottage valued at $250,000 from his great-uncle who owned a number of such buildings. The cottage was rented each year to college students who...
-
What is the adjustment that must be made to taxable income in computing alternative minimum taxable income for 2019, assuming that an individual purchased an office building on January 8 for $400,000?
-
Steven Spokesman, a high school teacher in - a local school district, was fired for publicly criticizing the local school district. The court ruled that Steven's freedom of speech had been violated...
-
On September 23, 2019, Mary Jones bought an annuity contract for $22,050 that will give her $125 a month for life, beginning October 30. Mary is 61 years old. Determine the exclusion ratio and the...
-
Assume the same facts as in Problem 31, except that in January 2020, because of a cost-of-living increase, Don's annuity payment was increased to $175 per month. Determine the amount of the 2020...
-
Every state has a law(s) regulating its own budgetary practices and those of its local governmental units. Check the law(s) of your state to determine the following: 1. For what funds must budgets be...
-
CdF2 (s) Cd+ (aq) + 2 F- (aq) 1. A saturated solution of CdF2 is prepared. The equilibrium in the solution is represented above. In the solution [Cd+] eq = 0.0585 M and [F-] eq = 0.117 M. a....
-
Identify the reagents you might use to achieve each of the following transformations: Br Br , En
-
Single bonds generally experience free rotation at room temperature: Nevertheless, the single bond shown below exhibits a large barrier to rotation. In other words, the energy of the system is...
-
Starting with acetylene as your only source of carbon atoms, identify how you would prepare each member of the following homologous series of aldehydes: a. Ethanal b. Propanal c. Butanal d. Pentanal
-
rewrite longer In January 2023, Apple announced that in 2024, it will bring back manufacturing in-house. Several factors may influence Apple's decision to insource certain operations or functions....
-
When the teacher gave an exercise after the lesson, the teacher found that students were not able recall what was explained. Explain, using any forgetting theory why this could have happened. Also...
-
For many astute investors, the most important variable of business plans is Blank______. Multiple choice question. the idea financial projections the environmental context the people

Study smarter with the SolutionInn App


