How would you prepare the following ketones by reaction of a Grignard reagent and a nitrile? (a)
Question:
How would you prepare the following ketones by reaction of a Grignard reagent and a nitrile?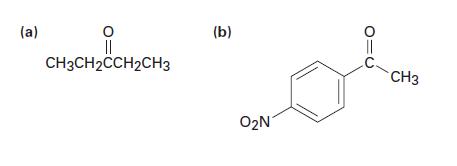
Transcribed Image Text:
(a) || CH3CH2CCH2CH3 (b) O₂N CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
To prepare ketones using a Grignard reagent and a nitrile you would follow these steps a CH3CH2CCH2C...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
2. What is the most important difference between generic software product development and custom software development? What might this mean in practice for users of generic software products? (2...
-
On 1 April 2019, Lisa borrowed $47204 at an interest rate of 2.37% from her employer, Solarcity Pty Ltd. This was lower than that offered to the public. Lisa uses the entire amount to replace the...
-
The Reformatsky reaction is an addition reaction in which an organozinc reagent is used instead of a Grignard reagent to attack the carbonyl group of an aldehyde or a ketone. Because the organozinc...
-
In Problems 1118, mentally solve each equation. 6x = -24
-
The contribution margin ratio of the Furniture Department at Glad's Mercantile is 0.75. The trace able fixed costs for the Furniture Department are estimated at $188,000 per year. Sales in the...
-
Your boss is considering the use of a new predictor. The base rate is high, the selection ratio is low, and the validity coefficient is high for the current predictor. What would you advise your boss...
-
What is the purpose of practice aids in forensic and valuation services?
-
Premiums Presented below are three independent situations. 1. Mar-quart Stamp Company records stamp service revenue and provides for the cost of redemptions in the year stamps are sold to licensees....
-
1 Problem 4 - A firm's optimal output choice (10 points) Consider a price taking firm, where the market price for the output is given by P. The firm's output choice is denote by Q. The firm has a...
-
Show the enolate ions you would obtain by deprotonation of the following carbonyl compounds: (a) 0 CH3CHCHCH (b) 0 CH3CCHCH3 (c) 0 CH3
-
Show the products of hydrolysis of the following esters: (a) 0 CH3 || | CH3COCHCH3 (b) COCH3
-
Solve the inequality. Then graph the solution set. (Some inequalities have no solution.) 1. x < 5 2. x 8 3. x / 2 > 1 4. x / 3 < 2 5. x - 5 < - 1 6. x - 7 < -5
-
In problem 49, when does the estate's taxable year end? Problem 49 John Henry died on May 1, 2019. When does his final 1040 have to be filed?
-
John Doe had earned commissions on life insurance sold prior to his death. The commissions of $20,000 were to be paid in five annual installments. The executor of his estate collected the first...
-
An estate with depreciable property may not do one of the following: a. Pay income to first-tier beneficiaries b. Pay income to second-tier beneficiaries c. Distribute capital losses on termination...
-
The following court cases have helped to clarify the rightful recipient of depreciation of property held by an estate or trust. Read the following judicial decisions and prepare a brief written...
-
When a trust terminates, there are, in most instances, terminating commissions that are charged by the fiduciary. The allocation of such expenses with respect to character of the income has spawned...
-
Compute the debit or credit, if any, to be made to the Budgetary fund balance for a countys General Fund under each of the following assumptions: a. Budgeted revenues and expenditures each are $...
-
We all experience emotions, but some people disguise their true feelings better than others. Do you think this is a helpful or harmful thing to do? Under what conditions do you think it would be most...
-
Consider the following two compounds: a) Identify which of these two compounds has greater resonance stabilization. b) Would you expect compound C (below) to have a resonance stabilization that is...
-
In each case below, identify the acid and the base. Then draw the curved arrows showing a proton transfer reaction. Draw the products of that proton transfer, and then predict the position of...
-
Identify the reagents you would use to accomplish each of the following transformations: Br . - Br Br En En
-
is it theoretically possible for the heat gain by cool water to exceed heat loss by warm water? Explain how your answer relates to the heat gained by the calorimeter, f in Part I in the previous...
-
A fully amortizing CPM mortgage loan is made for $100,000 at 6 percent interest for a 30 year term. Determine payments for each of the periods below if interest accrues: Monthly Quarterly Annually...
-
Ideally, a nation acquires funds for investment spending on plant and equipment by Blank______ income. Multiple choice question. generating saving borrowing financing

Study smarter with the SolutionInn App


