What alcohols might the following alkenes be made from? (a) CH3 CH3 (b) CH3CHCH=CHCHCHCH3
Question:
What alcohols might the following alkenes be made from?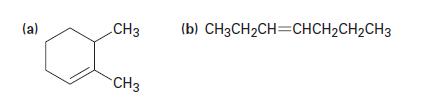
Transcribed Image Text:
(a) CH3 CH3 (b) CH3CH₂CH=CHCH₂CH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To determine the alcohols that the given alkenes might be made from we need to identify the possible ...View the full answer

Answered By

Surendar Kumaradevan
I have worked with both teachers and students to offer specialized help with everything from grammar and vocabulary to challenging problem-solving in a range of academic disciplines. For each student's specific needs, I can offer explanations, examples, and practice tasks that will help them better understand complex ideas and develop their skills.
I employ a range of techniques and resources in my engaged, interesting tutoring sessions to keep students motivated and on task. I have the tools necessary to offer students the support and direction they require in order to achieve, whether they need assistance with their homework, test preparation, or simply want to hone their skills in a particular subject area.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What alkenes might be used to prepare the following alcohols by hydroboration/oxidation? (a) CH3 (b) CHH (c) .CH- CHCHCH2CH2OH
-
What alkenes might the following alcohols have been prepared from? (b) (a) CH3CCH2CH2CH2CH3 C
-
What alkyl halides might the following alkenes have been madefrom? (b) C CH (a) (a) CH CHH2H2%3DCH2 "CH
-
In Problems 25 54, solve each system. Use any method you wish. 2xxy + y = 8 xy = 4
-
A firm has $ 1.5 million in sales, a Lerner index of 0.57, and a marginal cost of $ 50, and competes against 800 other firms in its relevant market. a. What price does this firm charge its customers?...
-
Consider the figure below. Similar to our discussion of Figure 9.1. Suppose that video is encoded at a fixed bit rate, and thus each video block contains video frames that are to be played out over...
-
Ormet Primary Aluminum Corporation, operated an aluminum smelter plant in Hannibal, Ohio. The facility ceased production in October 2013 in order to liquidate its assets after filing for bankruptcy...
-
Fiber Systems manufactures an optical switch that it uses in its final product. The switch has the following manufacturing costs per unit: Another company has offered to sell Fiber Systems the switch...
-
Following are the ledger balances of Titas Pvt. Ltd. as on the date 31 December, 2022. Prepare the Trial Balance using the following balances. Account Name Tk. Bank Overdraft 40,000 Cash 20,000...
-
What effect would the following changes have on the rate of the S N 1 reaction of tert-butyl alcohol with HBr? (a) The HBr concentration is tripled. (b) The HBr concentration is halved, and the...
-
What product would you expect to obtain from the S N 2 reaction of (S)-2 bromohexane with sodium acetate, CH 3 CO 2 Na? Show the stereochemistry of both product and reactant.
-
Distinguish between F.O.B. destination and F.O.B. shipping point.
-
A firm has a fixed cost of $100,000, and variable cost is $90,000 at an output of one. How much is marginal cost at an output of one? a) $10,000 b) $90,000 c) $100,000 d) $190,000 e) There is...
-
Parkinsons l aw is an example of _______. a) economies of scale b) diseconomies of scale c) Adam Smiths pin factory d) the firms search for its most profitable output.
-
A perfectly elastic supply curve is ______. a) a horizontal line b) a vertical line c) neither a horizontal nor a vertical line
-
When Kelly Ziegenfuss buys five units of a particular good or service, _______. a) she has no consumer surplus b) she has a consumer surplus c) there is no way of knowing whether she has a consumer...
-
When demand is relatively inelastic and supply is relatively elastic, the burden of a tax will be borne ______. a) mainly by sellers b) mainly by buyers c) equally between sellers and buyers d) it is...
-
What is collaboration? How can supply chains use collaboration to their advantage?
-
As water moves through the hydrologic cycle, water quality changes are common because of natural phenomena or anthropogenic pollution. Using Figure 11.1, describe how water-quality changes occur...
-
Identify whether each of the following reagents would be a strong nucleophile or a weak nucleophile, and also indicate whether it would be a strong base or a weak base: a.
-
We have seen that NaH is a strong base but a weak nucleophile. In contrast, lithium aluminum hydride (LAH) is a reagent that can serve as a source of nucleophilic hydride ion: In this case, LAH...
-
a) NaOH is a strong nucleophile and strong base. The substrate in this case is primary. Therefore, we expect S N 2 (giving the major product) and E2 (giving the minor product). b) NaSH is a strong...
-
3 Graph the line with slope passing through the point (-5, -2).
-
Sometimes we have incomplete information, or we make mistakes. Often people follow a set approach because they are afraid of trial and error. Part of critical thinking involves looking at opposing...
-
1. Deacribe how Ron Shaich uses the functions of management. 2. As CEO what are some of the major plans that Shaich has made for Panera Bread? what element of planning was he using here ?

Study smarter with the SolutionInn App


