What carbonyl compounds would you reduce to prepare the following alcohols? List all possibilities. (a) CH3 CH3CHCHCHCCHOH
Question:
What carbonyl compounds would you reduce to prepare the following alcohols? List all possibilities.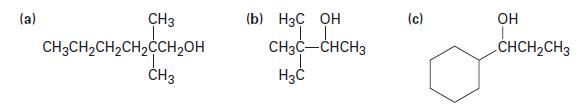
Transcribed Image Text:
(a) CH3 CH3CH₂CH₂CH₂CCH₂OH CH3 (b) H3C OH CH3C-CHCH3 H3C (c) OH CHCH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To prepare the alcohol CH3CH2CH2CH2CCH2OH you would need to reduce a carbonyl compo...View the full answer

Answered By

User l_1006857
I am a computer science professional with expertise in databases, AI programming, data structures and algorithms, and mathematics. With a strong background in these areas, I possess the knowledge and skills necessary to design and optimize database systems, develop intelligent algorithms and models, and solve complex computational problems. My proficiency in SQL, NoSQL, machine learning techniques, and mathematical concepts equips me to contribute to innovative projects and drive technological advancements.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What carbonyl compounds would you reduce to prepare the following alcohols? List all possibilities. (b) CH |(a) (c) H CHCH2CH2H2H2 C CHCH-CH
-
What carbonyl compounds might you start with to prepare the following compounds by Grignard reaction? List all possibilities. (a) 2-Methyl-2-propanol (b) 1-Ethylcyclohexanol (c) 3-Phenyl-3-pentanol...
-
Show what alcohols and carbonyl compounds give the following derivatives. (a) (b) (c) (d) (e) (f) CH CH,O OCH,CH CH O-CH CH3 CH3 CH-C H O-CH CH a,0 OX
-
Problems 113 122. The purpose of these problems is to keep the material fresh in your mind so that you are better prepared for later sections, a final exam, or subsequent courses such as calculus. X...
-
The French government announced plans to convert state- owned power firms EDF and GDF into separate limited companies that operate in geographically distinct markets. BBC News reported that Frances...
-
In CRC, which of the following generators (divisors) guarantees the detection of a single bit error? a. 101 b. 100 c. 1
-
On April 20, 1992, Daniel Hubbard (plaintiff), a potato farmer, and UTZ Quality Foods, Inc. (UTZ) (defendant), a potato chip manufacturer, entered an installment contract under which Hubbard agreed...
-
The income statement, balance sheets, and additional information for Video Phones, Inc., are provided. Additional Information for 2015: 1. Purchase investment in bonds for $115,000. 2. Sell land...
-
Ricardo works for Bank B and is talking about loans with a consumer. He provides the consumer with a general explanation regarding the basic qualifications of a loan. Although the consumer plans to...
-
Which ion in each of the following pairs is a better leaving group? (a) F - or Br - (b) Cl - or NH 2 - (c) OH - or I -
-
Describe the effects of the variables on both S N 2 and S N 1 reactions: (a) Substrate structure (b) Leaving group
-
Let n be a positive integer. For which n are the two infinite one-sided limits lim 1/x equal?
-
Compare and contrast problem analysis and root cause analysis. Under what conditions would you use problem analysis? Under what conditions would you use root-cause analysis?
-
When the supply of money falls, interest rates _________.
-
Suppose that the forward ask price for March 20 on euros is $0.9127 at the same time that the price of CME euro futures for delivery on March 20 is $0.9145. How could an arbitrageur profit from this...
-
What happens to interest rates when the demand for money rises? _______.
-
Colleges and universities need to stay current with technologies. Many campuses have adopted laptop programs, where students are expected to purchase or lease a particular model of laptop that will...
-
The annual report is considered by some to be the single most important printed document that companies produce. In recent years, annual reports have become large documents. They now include such...
-
Propose a reasonable mechanism for the following reaction. OH
-
The cyclopropenyl cation has a three-membered ring that contains a continuous system of overlapping p orbitals. This system contains a total of two Ï electrons. Using a Frost circle, draw an...
-
Arrange each set of isomeric alkenes in order of stability. a. b.
-
Consider the following two isomeric alkenes. The first isomer is a mono-substituted alkene, while the second isomer is a di-substituted alkene. We might expect the second isomer to be more stable,...
-
L In triangle ABC, if a = 12cm b = 9cm , and ZA = 60 , find using the Law of Sines. LB
-
From the top of a 100-meter high tower, the angle of depression to a car on the ground is 30 degrees. How far is the car from the base of the tower?
-
Prove that tan 2 ()+1=sec 2 ()

Study smarter with the SolutionInn App


