(a) Arrange the following esters in order of increasing reactivity toward glycinamide (the amide of the amino...
Question:
(a) Arrange the following esters in order of increasing reactivity toward glycinamide (the amide of the amino acid glycine), least reactive first, and explain your choice.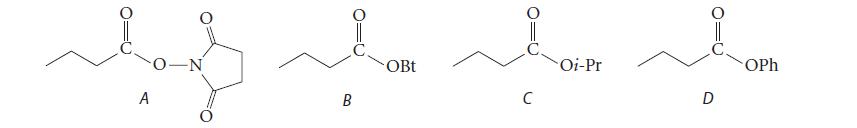
(b) Give the product of the reaction of glycinamide with B.
Transcribed Image Text:
A -N bom bom bom OBt Oi-Pr OPh B C D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a b The reactivity of the esters is related to the stability of the leaving group The more st...View the full answer

Answered By

MICHAEL KICHE
I was employed studypool for the first time in tutoring. I did well since most of my students and clients got the necessary information and knowledge requested for. I always submitted the answers in time and followed the correct formatting in answering eg MLA or APA format,
Again I worked with the writers bay where I did writing and got many clients whom we worked with so closely. They enjoyed every single service I delivered to them. My answers are always correct.
4.70+
13+ Reviews
52+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the following compounds in order of increasing reactivity toward HNO3 in H2SO4. (The references to equations will assist you with nomenclature.) (a) Chlorobenzene, benzene, nitrobenzene (b)...
-
a. List the following esters in order of decreasing reactivity in the first step of a nucleophilic acyl substitution reaction (formation of the tetrahedral intermediate): b. List the same esters in...
-
Rank the following dienes in order of increasing reactivity in a Diels-Alder reaction (1 = least reactive, 4 = most reactive). Briefly explain your ranking.
-
ABC Ltd prepares its financial statements to 31 October each year. Its trial balance at 31 October 2019 was as follows: Premises-cost Manufacturing plant-cost Office equipment-cost Accumulated...
-
You have the opportunity to invest $10,000 in one of two companies from a single industry. The only information you have is shown here. The word high refers to the top third of the industry; average...
-
Bradbury Corporation turns its inventory five times each year, has an average payment period of 25 days, and has an average collection period of 32 days. The firms annual sales are $3.6 billion, its...
-
Match the measures of worth in the first column with the appropriate unit of measure that results from the analysis. Measure of Worth (a) Annual Worth (b) External Rate of Return (c) Future Worth (d)...
-
For 2012, Selene Company initiated a sales promotion campaign that included the expenditure of an additional $25,000 for advertising. At the end of the year, Scott Brown, the president, is presented...
-
What are some reasons why understanding the organization of the Internal Revenue Code may prove useful in the accounting world no matter what your job title is ?
-
What average yield per amino acid would be required to synthesize a protein containing 100 amino acids in 50% overall yield?
-
Calculate the average yield of each of the 369 steps in the synthesis of ribonuclease by the solid-phase method discussed, assuming the reported overall yield of 17%.
-
Use the Target Corporations Form 10-K to answer the following questions related to Targets 2012 fiscal year (year ended February 2, 2013). Targets Form 10-K is available on the companys website or...
-
Which factor is generally given the most weight in apportioning business income between states? Which factor is generally given the most weight in apportioning business income between states?...
-
What is the room departments income? TOTAL REVENUE Rooms Revenue 100.00% 2,500,000 Payroll and Related Expenses 500,000 Other Expenses 200,000 Department Income Food-Revenue 1,000,000 Cost of Sales...
-
For what minimum period of time must a tax return preparer make a client taxpayer's e-file signature authorization available to the IRS?
-
Sales $ 397,596 $ 261,576 $ 208,427 $ 149,410 $ 111,500 Cost of goods sold 209,250 137,520 111,682 79,143 57,980 Accounts receivable 19,283 15,302 14,256 8,711 7,627 Compute trend percents for the...
-
Use the link below to access the IRS Publication 17 website: https://www.irs.gov/publications/p17Links to an external site. Discuss the following: 1. What is the purpose of IRS Publication 17? 2. How...
-
Your friend, Drew, is a strong personality. When you explain the planning control cycle, Drew balks at the entire notion. He says, I know precisely what I want to do. Only indecisive people look back...
-
A simple random sample of 220 university students were asked what pasta they usually order and with which sauce. The preferences of these respondents are summarised below: Sauce Bolognese Pasta...
-
Which of the following compounds could be resolved into enantiomers at room temperature? Explain. (a) (b) CH2CH CH3)2 CH CH CH3 CH3
-
(a) When fumarate reacts with D2O in the presence of the enzyme fumarase only one stereoisomer of deuterated malate is formed, as shown in Fig. P7 .52.Is this a syn- or an ant I - addition ? Explain....
-
Give the structure and stereochemistry of all products formed in each of the following reactions. (a) (b) trans-3-pentene Br H2O excess (solvent) ,o.roH cis-3-hexene BD3 solvent)
-
1. Tobi Vail is a ski instructor and has an annual gross pay of $19,250. His exemptions total $1500. The state tax on the first $2500 is 1.5% and amounts over $2500 is 3%. What is the state income...
-
For a business plan of a bakery: Make the operational marketing to introduce the marketing mix 4Ps: In Promotion we have to state the 4 elements -advertisement -sales promotion -public relationship...
-
Write an explicit equation for the following table of values: X Y 0123456 10 60 2 360 3 2160 12960 7 77760 466560 2799360

Study smarter with the SolutionInn App


