(a) Explain why the SH stretching absorption in the IR spectrum of a thiol is less intense...
Question:
(a) Explain why the S—H stretching absorption in the IR spectrum of a thiol is less intense and occurs at lower frequency (2550 cm–1) than the O—H stretching absorption of an alcohol.
(b) Is the wavenumber difference between O—H and S—H absorptions caused primarily by the greater mass of sulfur or by the relative strengths of the two bonds? Explain how you know.
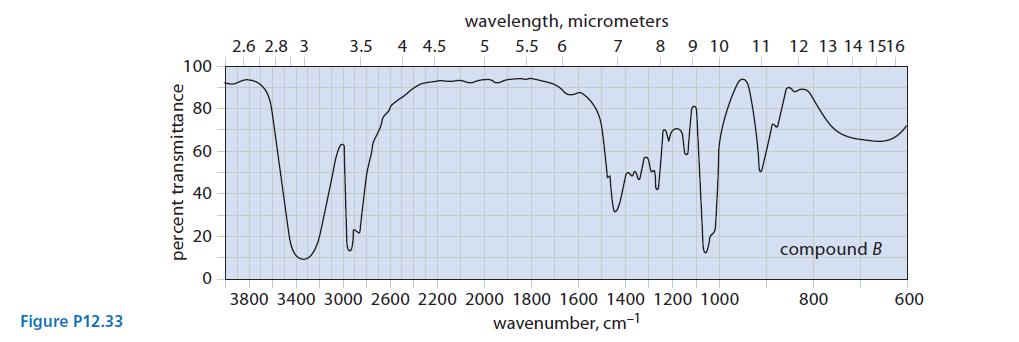
(c) Two unlabeled bottles, A and B, contain liquids. Laboratory notes suggest that one compound is (HSCH2CH2)2O and the other is (HOCH2CH2)2S.
The IR spectra of the two compounds are given in Fig. P12.33. Identify A and B and explain your choice.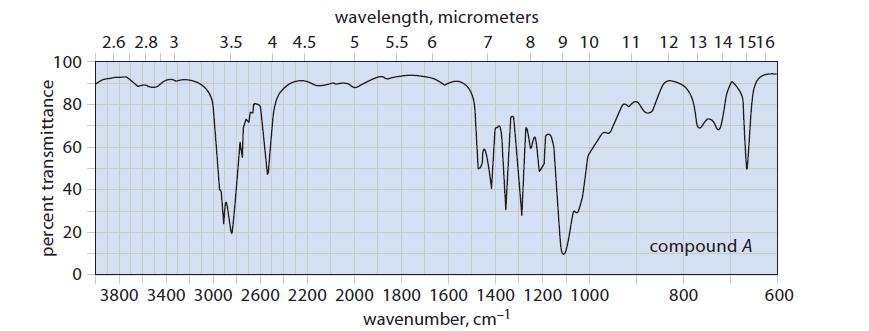
Transcribed Image Text:
100 percent transmittance 80 60 40 20 0 2.6 2.8 3 wavelength, micrometers 3.5 4 4.5 5 5.5 6 7 8 9 10 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm-¹ 11 12 13 14 1516 compound A 800 600
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a b c The bond dipole of the SH bond is much less than that of the OH bond because the electroneg ativities of sulfur and hydrogen differ less than do ...View the full answer

Answered By

Hassan Imtiaz
The following are details of my Professional Experience. Responsibilities Eight years of demanding teaching experience in the field of finance and business studies at Master’s Level. Completion of the given tasks within given time with quality and efficiency. Marketing professional with practical experience in and solid understanding of a diverse range of management applications, including market analysis, sales and marketing, team building and quality assurance. I have excellent skills to approach deal and sustain corporate clients / customers by demonstrating not only extraordinary communication and interpersonal skills but also high caliber presentation, negotiation and closing skills. Manage and follow up the day-to-day activities. Manage and co-ordinate the inventories. Fulfillment of all the tasks assigned.
The following are details of my Areas of Effectiveness. Finance 1. Corporate Finance 2. Advanced Corporate Finance 3. Management of Financial Institutions 4. International Financial Management 5. Investments 6. Fixed Income 7. Real Estate Investment 8. Entrepreneurial Finance 9. Derivatives 10. Alternative Investments 11. Portfolio Management 12. Financial Statement Analysis And Reporting (US GAAP & IFRS) 13. International Financial Markets 14. Public Finance 15. Personal finance 16. Real estate 17. Financial Planning Quantitative Analysis 1. Time Value Of Money 2. Statistics 3. Probability Distribution 4. Business Statistics 5. Statistical Theory and Methods Economics 1. Principles of Economics 2. Economic Theory 3. Microeconomic Principles 4. Macroeconomic Principles 5. International Monetary Economics 6. Money and Banking 7. Financial Economics 8. Population Economics 9. Behavioral Economics International Business 1. Ethics 2. Business Ethics 3. An introduction to business studies 4. Organization & Management 5. Legal Environment of Business 6. Information Systems in Organizations 7. Operations Management 8. Global Business Policies 9. Industrial Organization 10. Business Strategy 11. Information Management and Technology 12. Company Structure and Organizational Management Accounting & Auditing 1. Financial Accounting 2. Managerial Accounting 3. Accounting for strategy implementation 4. Financial accounting 5. Introduction to bookkeeping and accounting Marketing 1. Marketing Management 2. Professional Development Strategies 3. Business Communications 4. Business planning 5. Commerce & Technology Human resource management 1. General Management 2. Conflict management 3. Leadership 4. Organizational Leadership 5. Supply Chain Management 6. Law 7. Corporate Strategy Creative Writing 1. Analytical Reading & Writing Other Expertise 1. Risk Management 2. Entrepreneurship 3. Management science 4. Organizational behavior 5. Project management 6. Financial Analysis, Research & Companies Valuation 7. And any kind of Excel Queries
4.80+
150+ Reviews
230+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
Quiz# 5 (Q) The following are estimates for four risk assets (A,B,C,D). The portfolio P is an equal weighted portfolio of the four risk assets. Stock A Stock B Stock C Stock D Portfolio P weight 0.25...
-
What is the purpose of independent verification of performance?
-
In 150 words or fewer, list each of the four perspectives of the balanced scorecard. Give an example of one KPI from each of the perspectives and explain what measure the KPI provides for a retailing...
-
A company recognizes a lease as a capital lease when a. the lease term is less than 75% of the estimated useful life of the leased asset. b. the lease has no option to purchase the asset at the end...
-
The Atlantic Refinery Corporation ( ARC) is a public company headquartered in St. Johns, Newfoundland. On 31 December 20X5, the post- closing trial balance included the following accounts ( in...
-
Question 2 (2 points): Texarkana decided to retire its bonds, and made a gain on retirement of these bonds of $5,270. These bonds had a face value of $100,000, a 'premium on bond payable' at the...
-
Suggest structures for the following neutral molecules commonly lost in mass spectral fragmentation. (a) Mass = 28 from a compound containing only C and H (b) Mass = 18 from a compound containing C,...
-
Explain why a nitro compound has two NO stretching vibrations. (These typically occur at about 1370 and 1550 cm 1 .) R-N : :0: R-N :
-
Microsoft has embarked on a joint venture with Sony to develop an online alternative to the telephone. What type of offensive market strategy best describes this joint venture, and what are the...
-
Assuming 10 seconds is enough time for all of the tasks to finish, what statements about the following program are correct? (Choose two.) A. The code does not compile. B. The incrementBy10() method...
-
Am I aware of the basic framework of a strategic communication plan?
-
A sorority has 35 members, 25 of whom are full members and 10 of whom are pledges. Two persons are selected at random from the membership list of the sorority. Find the requested probabilities. a....
-
What are the required properties of molding sands? Classify the molding sand.
-
What are the various types of patterns? Explain them with their uses.
-
How are budgets used as levers of control?
-
Wimot Trucking Corporation uses the units-of-production depreciation method because units-of-production best measures wear and tear on the trucks. Consider these facts about one Mack truck in the...
-
1-Phenyl-2-butene has an ultraviolet absorption at max = 208 nm ( = 8000). On treatment with a small amount of strong acid, isomerization occurs and a new substance with max = 250 nm ( = 15,800) is...
-
What is the structure of a hydrocarbon that has M + = 120 in its mass spectrum and has the following 1 H NMR spectrum? 7.25 (5 H, broad singlet); 2.90 (1 H, septet, J = 7 Hz); 1.22 (6 H, doublet,...
-
Propose structures for compounds that fit the following descriptions: (a) C 10 H 14 H NMR: 7.18 (4 H, broad singlet); 2.70 (4 H, quartet, J = 7 Hz); 1.20 (6 H, triplet, J = 7 Hz) IR: 745 cm 1 (b)...
-
Two antennas located at points A and B are broadcasting radio waves of frequency 94.0 MHz, perfectly in phase with each other. The two antennas are separated by a distance d= 9.30 m. An observer, P,...
-
A group of white high school students from Dallas's Mockingbird High school were visiting Texas A&M University as part of their schools program "Road to College" program. These students attended a...
-
Arturo took out $38,000 in student loans at 4.25% interest. The standard repayment plan is to repay the loans in 10 years with equal monthly payments at the end of each month. (copyrighted exam...

Study smarter with the SolutionInn App


