Consider the detailed structure of the semiquinone? QH shown in Eq. 18.79a. (a) There are two possible
Question:
Consider the detailed structure of the semiquinone? QH shown in Eq. 18.79a.
(a) There are two possible structures for this semiquinone; draw them both.
(b) Show the resonance structures for either of the structures you gave in part (a).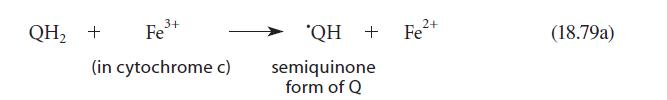
Transcribed Image Text:
QH₂ + 3+ Fe³+ (in cytochrome c) 'QH + Fe²+ semiquinone form of Q (18.79a)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a CH3O CH30 0 CH3 H 10 or CH3O ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
There are two possible structures of XeF2Cl2, where Xe is the central atom. Draw them, and describe how measurements of dipole moments might be used to distinguish among them.
-
Consider the following reactions. For parts b-d, reference Exercise 58. a. When C5H12 is reacted with Cl2(g) in the presence of ultraviolet light, four different monochlorination products form. What...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Show that an emission tax and an absolute emission standard are equivalent instruments to regulate a polluting monopolist if and only if the standard is binding.
-
How fast must you be moving toward a red light ( = 650 nm) for it to appear green ( = 525 nm)?
-
Anna Waldheim was seriously injured in an industrial accident. She sued the responsible parties and was awarded a judgment of $2,000,000. Today, she and her attorney are attending a settlement...
-
What skills might be useful in a cross-functional team that is responsible for designing a set-top box?
-
Green Valley Company prepared the following trial balance at the end of its first year of operations ending December 31, 2014. To simplify the case, the amounts given are in thousands of dollars....
-
Name the financial statement where each of the following will appear: (IS) Income Statement; (BS) Balance Sheet; (SCF) Statement of Cash Flows; (N) None. a. Book value of equipment purchased five...
-
Draw the important resonance structures of the radicals formed when each of the following reacts with R?, a general free radical. (a) Vitamin E (b) BHT
-
(a) Using the fishhook notation, derive the important resonance structures of the vitamin C-derived radical in Eq. 18.81b. (b) In the laboratory, the radical derived from vitamin E can react with a...
-
How has Theo Chocolate incorporated its model of philanthropy and social responsibility into a successful business concept?
-
The following graph shows inflation and unemployment rates for Canada for the period between 1970 and 2012. Does this graph show evidence in favor of the Phillips curve? 14 12 10 8 6 4 1 O 2 Canada...
-
Assuming that Okuns law is given by U U n = 0.75 (Y Y P ) and that the Phillips curve is given by = e - 0.6 (U U n ) + , a) Obtain the short-run aggregate supply curve if expectations are...
-
The following table shows unemployment and inflation rates for Canada during the 19721982 period: a) Plot Canadas unemployment rates during this period. On the same graph, draw a horizontal line at...
-
What would happen to revenue from seignorage if the inflation rate was very high? check Equation 8 and assume a quickly rising price level. M/P = (M/P) (8)
-
From a risk perspective, how is a forward contract different from a futures contract?
-
According to the American Medical Association, about 36% of all U.S. physicians under the age of 35 are women. Your company has just hired eight physicians under the age of 35 and none is a woman. If...
-
Imagine that your best friend knows you are taking a psychology course and wonders what psychology is all about. How would you define psychology for your friend? Write an essay on the discipline of...
-
Draw the most stable conformation of: (a) ethylcyclohexane (b) 3-isopropyl-1,1-dimethylcyclohexane (c) cis-1-tert-butyl-4-isopropylcyclohexane
-
(a) Draw both chair conformations of cis-1,4-dimethylcyclohexane, and determine which conformer is more stable. (b) Repeat for the trans isomer. (c) Predict which isomer (cis or trans) is more stable.
-
Use your results from Problem 3-25 to complete the following table. Each entry shows the positions of two groups arranged as shown. For example, two groups that are trans on adjacent carbons...
-
How can the principles of servant leadership be applied to improve organizational effectiveness? Explain
-
Look for news articles/research studies written and conducted across the world about the sociocultural impact of the "Tinder" organization/company. Make an analysis on the gathered information in...
-
a) Resolve x + 28k (x-2k)(x+3k) into partial fractions, where k is a constant. b) The area under the curve y=ex +3 between x = In 2 and x = ln 4 is rotated 360 about the x-axis. Find the volume...

Study smarter with the SolutionInn App


