Each of the following compounds can be resolved into enantiomers. Explain why each is chiral, and why
Question:
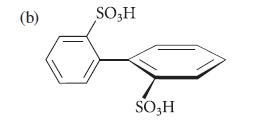
Each of the following compounds can be resolved into enantiomers. Explain why each is chiral, and why compound (b) racemizes when it is heated.![(a) hexahelicene [a] =3700 degrees mL g- dm-](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/6/7/0/302656d6d9ee8e731701670301706.jpg)
Transcribed Image Text:
(a) hexahelicene [a] =3700 degrees mL g-¹ dm-¹
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a The two terminal rings of hexahelicene cannot lie in the same plane because if they did their hydr...View the full answer

Answered By

Somenath Mukhopadhyay
After working for almost twenty years in the corporate software job, i have started training students in software. I have been doing it for the last 5 years. Among my students there are many kid-students and i am proud to say that these kid-students are doing really good in programming. Some kid-students are as young as 7-8 years of age and they explore quite advanced software subjects. Many of these students have their Google Play store profile. I teach them Java, Android, UML, Design Pattern, Scratch and many more. Here are the Google Play store profiles of some of my kid-students:
- https://play.google.com/store/apps/developer?id=Ridit+Mukhopadhyay&hl=en-GB
- https://play.google.com/store/apps/developer?id=Srijan+Sengupta&hl=en-GB
- https://play.google.com/store/apps/developer?id=ShreyasCoder&hl=en-GB
- https://play.google.com/store/apps/developer?id=Shankhadeep+Sarkar
Here are some of the videos of my kid-students taking software classes -
- https://youtu.be/NBTN9vfE9sE
- https://youtu.be/Ku_jJQTbTD4
- https://youtu.be/vmOf81W0414
- https://youtu.be/L0hgtXVJFKc
Here is the video of an impromptu Q & A session in my classroom
- https://youtu.be/GHavJXDHBQQ
- Here is my technical blog for advance learners:
http://som-itsolutions.blogspot.com/
I believe in hands-on knowledge and hence i stress on learning by doing. Come and enjoy the joy of learning.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds can be resolved into enantiomers at room temperature? Explain. (a) Z: (c) HC- CH3 CH3 CH3 (b) CHCH3 T CH3 CH3 (d) CHCH3 - CH3 H
-
Compound A, C8H11NO, smells as if it might have been isolated from an extract of dirty socks. This compound can be resolved into enantiomers and it dissolves in 5% aqueous HCl. Oxidation of A with...
-
Explain why compound A in Fig. P6.39 can be resolved into enantiomers but compound B cannot. CH3 1. PhN#CH_CH=CH, CI CH,Ph Figure P6.39 A CH3 T PhNCH,CH=CH2 B
-
What is the effect of a viscosity (competence) difference between strain markers and the matrix?
-
Derive cost of goods sold in data in the statement of cash flows The section showing cash flow from operations, using the indirect method, for Ann Taylor Stores reported an increase in inventories of...
-
Develop a brief answer to each of the following questions. 1. When a company has net income, what happens to its assets and/or to its liabilities? 2. Is accrual accounting more closely related to a...
-
You and your uncle are fishing. You are using 22-N fishing line, which means the line can support that much tension without breaking. Your uncle challenges you to catch the biggest fish you can land...
-
Compute the specified ratios using Faustin Companys balance sheet for 2014. Assets Cash .............. $ 18,000 Marketable securities ......... 12,000 Accounts receivable ........ 25,000 Inventory...
-
Brighton Bookstores has two divisions: books and electronics. The electronics division had another great year in 2024 with net sales of $9.1 million, cost of goods sold of $5.55 million, operating...
-
Given that anisole (methoxybenzene) protonates primarily on oxygen in concentrated H 2 SO 4 , explain why 1,3,5-trimethoxybenzene protonates primarily on a carbon of the ring. As part of your...
-
A DielsAlder reaction of 2,5-dimethylfuran and maleic anhydride gives a compound A that undergoes acidcatalyzed dehydration to give 3,6-dimethyl phthalic anhydride (see Fig. P16.65). (a) Deduce the...
-
Eastwood Manufacturing bought three used machines in a $216,000 lump-sum purchase. An independent appraiser valued the machines as shown in the table. Machine No. Appraised Value 1 ............. $...
-
Return to the bicycle manufacturer NatBike in Exercise 6. Assume that the plant has a capacity of 20,000 bicycles. If additional capacity can be added at a cost of $25 per bicycle, how should NatBike...
-
A married couple with two children has earned income of $30,000 with no withholding. Assume they have a taxable income of $100, they paid $2,295 in FICA taxes, and they will receive an earned income...
-
A married couple with two children has $16,000 of earned income. What is the amount of their child and family tax credit?
-
Sara's only source of income was wages of $6,000. She and her daughter live with her parents. The daughter is claimed as a dependent by the father. Is Sara eligible for the earned income credit?
-
What is a tax preference item?
-
Executives often spend so many hours in meetings that they have relatively little time to manage their individual areas of operation.What is the difference in mean time spent in meetings by...
-
In Exercises 1-2, rewrite each verbal statement as an equation. Then decide whether the statement is true or false. Justify your answer. 1. The logarithm of the difference of two numbers is equal to...
-
What product is obtained in each case when 3-hexyne is treated in each of the following ways? With H2 over Pd/C and quinoline and the product of that reaction with D2 over Pd/C
-
(a) Using the pKa values of the hydrocarbons and ammonia, estimate the equilibrium constant for (1) the reaction in Eq. 14.22 and (2) the analogous reaction of an alkane with amide ion. Eq. 14.22 (b)...
-
Give the structures of the products in each of the following reactions. CH,Ca C: Na+ + CH,CH,-| - Br( CH2)-Br + HC C: Na+ (excess) -
-
Crane Co. has annual obligations of $30 toward interest and principal and is forecasting a cash flow of either $25 (Scenario I) or $50 (Scenario 2) in the coming year. What will the payment to...
-
Write a Python script to simulate Monte Carlo method using numpy library.
-
Access the IASB Web site IFRS - Resources for students. You can also use other sources to answer the following. a. Explain how the IFRS board develops IFRS Standards. b. Summarize the main features...

Study smarter with the SolutionInn App


