Explain why compound A in Fig. P6.39 can be resolved into enantiomers but compound B cannot. CH3
Question:
Explain why compound A in Fig. P6.39 can be resolved into enantiomers but compound B cannot.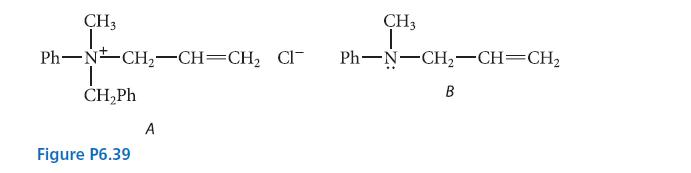
Transcribed Image Text:
CH3 1. Ph−N#CH_–CH=CH, CI CH,Ph Figure P6.39 A CH3 T Ph–N–CH,–CH=CH2 B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Compound B can undergo amine inversion which rapidly int...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Compound A, C8H11NO, smells as if it might have been isolated from an extract of dirty socks. This compound can be resolved into enantiomers and it dissolves in 5% aqueous HCl. Oxidation of A with...
-
Which of the following compounds can be resolved into enantiomers at room temperature? Explain. (a) Z: (c) HC- CH3 CH3 CH3 (b) CHCH3 T CH3 CH3 (d) CHCH3 - CH3 H
-
Which of the amines listed next can be resolved into enantiomers? In each case, explain why interconversion of the enantiomers would or would not take place. (a) cis-2-methylcyclohexanamine (b)...
-
To some U.S. manufacturers and labor unions, a cheap yuan value gives China's advantage in the global economy. bankers exports importers subsidies an unfair
-
McElveen served seven months and Milligan nine months. Both were ordered to pay $13,000 in restitution. Milligan lost his job on the space shuttle (and along with it, his health benefits). What...
-
Assume the same facts as in problem 98, except that Jinglings royalty income is from investments located in Japan. Identify the tax issue(s) posed by the facts presented. Determine the possible tax...
-
Petitioner Christy Brzonkala met respondents Antonio Morrison and James Crawford at a campus party at Virginia Polytechnic Institute (Virginia Tech), where they were all students. At the party, the...
-
Scofield Financial Co. is a regional insurance company that began operations on January 1, 2014. The following transactions relate to trading securities acquired by Scofield Financial Co., which has...
-
C Av > == Paragraph I LZ Question > |-- > 17 Styles 17 Seled Editing The Operations Manager of Toshiba's laptop manufacturing plant is about to prepare her annual report to the Board of Directors....
-
Construct sawhorse and Newman projections (Sec. 2.3A) of the three staggered conformations of 2-methylbutane (isopentane) that result from rotation about the C2C3 bond. (a) Identify the conformations...
-
Draw the structures of all compounds with the formula C 6 H 12 Cl 2 that can exist as meso compounds. Indicate how many meso compounds are possible for each structure.
-
Write a MATLAB program that will design a PI controller assuming a second-order approximation as follows: a. Allow the user to input from the keyboard the desired percent overshoot b. Design a PI...
-
In May 2004, Sodexho USA, the foodservice provider at Northwestern University, kicked off its second annual Global Chef program, a month-long international culinary residency that brings Sodexho...
-
Uality Inc. acquires Rekless Company for $100 million in cash at the beginning of its accounting year, and records the acquisition as a merger. At the date of acquisition, Rekless' reported net...
-
Currency transaction reports stemming from non-gaming areas of resorts are submitted by way of which form? Currency transaction reports stemming from the casino are submitted by way of which form?...
-
Using the DuPont analysis, where does Timberland's ROE come from? Does it come from profitability, asset management or leverage? Explain
-
Rental details from operation of two separate rental properties: Property 1 Property 2 Property 3 Gross rental revenue . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $62,000 $52,000...
-
Distinguish between explicit and implicit costs, giving examples of each. What are some explicit and implicit costs of attending college? Why does the economist classify normal profit as a cost? Is...
-
Briefly describe the following types of group life insurance plans: a. Group term life insurance b. Group accidental death and dismemberment insurance (AD&D) c. Group universal life insurance d....
-
What product(s) would you expect to obtain from the followingreactions? (b) r. 1. HNO3, H2SO4 2. Fe, H30* "C Br H2/Pd NO2 (d) CI. (c) CH3CH2CH2CI MO 2 AICI3
-
Predict the major product(s) of the followingreactions: (b) (a) CH3CH2COCI AICI3 CH3CH2CI AICI3 (c) (d) N(CH2CH3}2 CO2H HNO3 so3 H2SO4 H2SO,
-
Aromatic iodination can be carried out with a number of reagents, including iodine mono chloride, ICI. What is the direction of polarization of ICI? Propose a mechanism for the iodination of an...
-
Describe the type of the object x in the following declarations in English (for example: int **x: x is a pointer to a pointer to integer) int *x [10]; int (*x) (); int (**[]));
-
The complete Fourier series for a function F(x) is given by 00 ) = a+ a cos(nx)+2b,sin(nx) F(x)= n=1 n=1 Write a program that uses counter-controlled iteration to compute an approximation of F(x) by...
-
Use Minitab to answer the following questions 3. An experiment was run to investigate the influence of DC bias voltage on the amount silicon dioxide etched from a wafer in a plasma etch process....

Study smarter with the SolutionInn App


