Which of the following compounds can be resolved into enantiomers at room temperature? Explain. (a) Z: (c)
Question:
Which of the following compounds can be resolved into enantiomers at room temperature? Explain.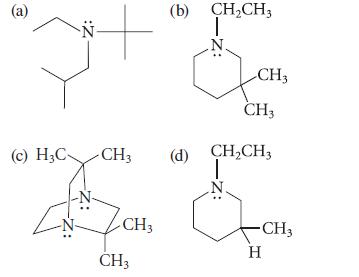
Transcribed Image Text:
(a) Z: (c) H₂C- CH3 CH3 CH3 (b) CH₂CH3 T CH3 CH3 (d) CH₂CH3 ☆ - CH3 H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a The only stereocenter in the molecule is the nitrogen which rapidly u...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds could be resolved into enantiomers at room temperature? Explain. (a) (b) CH2CH CH3)2 CH CH CH3 CH3
-
Which of the following compounds could in principle be resolved into enantiomers at very low temperatures? Explain. (a) Propane (b) 2,2,3,3, -tetramethylbutane
-
Which of the following compounds could in principle be resolved into enantiomers at very low temperatures? Explain. (a) Propane (b) 2,3-dimethylbutane (c) 2,2,3,3-tetramethylbutane
-
Freight and other handling charges on goods out on consignment are part of the cost of goods consigned. What is its appropriate account title in the income statement prepared by the consignor?...
-
Discuss trends impacting marketing and the implications of these trends on how marketers deliver value to customers.
-
The Windwalker Company has collected the following data for one of its product lines: total production costs, $300,000; total selling, general, and administrative expenses, $112,600; desired profit,...
-
A European recession and the U.S. economy a. In 2014, European Union spending on U.S. goods accounted for \(18 \%\) of U.S. exports (see Table 17-2), and U.S. exports amounted to \(15 \%\) of U.S....
-
Listed below are items that are commonly accounted for differently for financial reporting purposes than they are for tax purposes. Instructions For each item below, indicate whether it involves: 1....
-
Criticism of the World Bank is generally on a diverse range of issues but they generally centre around concern about the approaches adopted by the World Bank in formulating their policies, and the...
-
Explain why 1-methylaziridine undergoes amine inversion much more slowly than 1-methylpyrrolidine. (What are the hybridization and bond angles at nitrogen in the transition state for inversion?)...
-
Draw a chair conformation for (S)-3-methylpiperidine showing the sp 3 orbital that contains the nitrogen unshared electron pair. How many chair conformations of this compound are in rapid...
-
What are the benefits of partnering?
-
Farah Snack Co. has earnings after taxes of $150,000. Interest expense for the year was $20,000; preferred dividends paid were $20,000; and common dividends paid were $30,000. Taxes were $22,500. The...
-
Comparison between old Operating Systems and contemporary Operating System as well as comparison between different types of Operating systems
-
what is the difference between Network operating System and operating system? which software can i say is Network operating System?
-
Christopher and Simone attended a 'financial well-being' seminar offered by Ladkins Financial around six months ago. Some of the tips they took away from the seminar were related to reviewing their...
-
Royal Dutch Shell (RDSA) will reward patient shareholders with a massive stock buyback even as it missed profit estimates. while ConocoPhillips (COP) narrowly beat earnings views as oil prices...
-
Using an allowable shear stress of 50 MPa, determine the power that can be transmitted at 2000 rpm through a shaft with a 30-mm diameter.
-
The maximum pressure that can be developed for a certain fluid power cylinder is 15.0 MPa. Compute the required diameter for the piston if the cylinder must exert a force of 30 kN.
-
Show the products, including stereo chemistry, of these SN1 reactions: a) CHCH Ph CH3 + CHOH HC b) CHCH H CH, -CH-C-CI+ CHCOH CH
-
Explain whether this reaction would follow the SN1 or the SN2 mechanism and then explain which reaction is faster: CI a) CHCH + OH HO CHOH CH3 b) CHC-Br + CHCO CH, O-SOCH3 or CHCH CH CO,H or + OH CH3...
-
(a) Show all the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of electrons in each step of the mechanism. (b) Show a free energy versus reaction...
-
d) 3) Use the given information to calculate K for the following reaction and then determine the composition of the reaction mixture: N2(g)+O2(g) 2NO(g) a) 1.6x103; the equilibrium mixture contains...
-
Bearcat, Inc. had actual manufacturing overhead costs of $ 1 , 1 6 0 , 0 0 0 and a predetermined overhead rate of $ 4 . 0 0 per machine hour.Bearcat, Inc. worked 2 9 0 , 0 0 0 machine hours during...
-
Calculate the change in pH when 9.00 mL of 0.100 M HClaq) is added to 100.0 mL of a buffer solution that is 0.100 M inNH: (aq) and 0.100 M in NH, Claq). Consult the table oflipnization constants as...

Study smarter with the SolutionInn App


