From the data in Fig. 8.1, tell which bonds have the greater amount of p character (Secs.
Question:
From the data in Fig. 8.1, tell which bonds have the greater amount of p character (Secs. 1.9B and 6.9B): C—O bonds or C—S bonds. Explain.
Fig. 8.1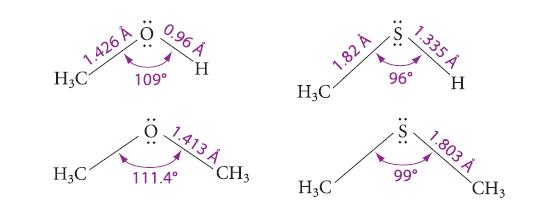
Transcribed Image Text:
1.426 Á H₂C H₂C 109° 0.96 Å H 1.413 A 111.4° CH3 1.82 A H3C1 H₂C 1.335 Å 96° H 99⁰ 1.803 Å CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
If bonds to an atom involved only its p orbitals then the bond ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Emily Jackson (Social Security number 765-12-4326) and James Stewart (Social Security number 466-74-9932) are partners in a partnership that owns and operates a barber shop. The partnership's first...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
The labor force participation rate is increasing in Japan because... a) there are fewer unemployed people b) the working age population is increasing c) retirement ages are increasing d) more women...
-
Jean-Franois, a French wine exporter, sues Bob Joe, a Texas importer, claiming that Bob Joe owes him $2 million for wine. Jean-Franois takes the witness stand to describe how the contract was...
-
Explain the pay-as-you-go system.
-
Determine the number of ideal stages required in Example 7.4 if the solvent rate used is twice the minimum. Data From Example 7.4:- A solution of acetic acid (C) in water (A) is to be extracted using...
-
Austin, Inc., acquired 10 percent of McKenzie Corporation on January 1, 2014, for $210,000 although McKenzies book value on that date was $1,700,000. McKenzie held land that was undervalued by...
-
Accounting information systems collect and process information from transactions and then organizes it into useful information for company management and typically include the general journal, which...
-
(a) The dipole moment of acetaldehyde is 2.7 D and that of propene is 0.5 D. Even though they have about the same molecular mass, they differ in boiling point by about 68C (47C and 121C). Which has...
-
(a) A chemist used the name 3-butyl-1,4-dioxane in a paper. Although the name unambiguously describes a structure, what should the name have been? Explain. (b) Give the structure of 2-butoxyethanol,...
-
Given three batteries with voltages of 1.0 V, 3.0 V, and 12 V, what are the minimum and maximum voltages that could be achieved by connecting them in series?
-
Consumers looking for other ways to get what they need in tighter times like the movement away from traditional bookstores to tablets is tied to what challenge or opportunity facing marketers today?
-
Discuss the significant trade-off between two competing objectives in hospital information systems. What are the competing objectives? What is the nature of the trade-off? Be detailed and thorough in...
-
Pushpinder is riding on a crowded bus. He has no place to sit, but instead is holding onto one of the overhead straps. Darren is standing next to Pushpinder, also holding onto a strap. Suddenly a car...
-
explain how Tesla apply the 5 performance objectives to operations management.
-
What kind of expenses a greengrocer may incur, and how would you relate the law of diminishing returns to his shop? Identify economies of scale in this enterprise. Quantify the length of the...
-
Refer to Table 11W.1 and suppose the price of new product C is $2 instead of $4. TABLE 11W.1 Utility Maximization with the Introduction of a New Product (Income = $10)* How does this affect the...
-
Suppose the spot and six-month forward rates on the Norwegian krone are Kr 5.78 and Kr 5.86, respectively. The annual risk-free rate in the United States is 3.8 percent, and the annual risk-free rate...
-
Show equations to account for the major fragment ions that occur at the indicated m/z for thesecompounds: CH,CI b) CH3CH2CH,CH2OH nal: 31 miz 91 c) CH;CH,CCH,CH,CH,CH3 mtz 85, 72, 57
-
A student wishes to record the UV spectrum of trans-stilbene, which has max = 308nm ( = 25,000), what concentration should be prepared if the desired absorbance is 0.5 at the maximum?
-
Indicate the types of transitions responsible for the absorptions of these compounds: Apax = 252 nm (e = 20,000) Aas - 325 nm ( = 180) a) A mux = 235 nm (e = 19,000) b) c) Amax = 299 nm ( = 20) d)...
-
An AC generator is connected to a 15.00 resistor. The output voltage of the generator is given by the function Av = (180 V)sin[(80 rad/s)t]. (a) From the equation for the output voltage, what is the...
-
1) Calculate the drift velocity v_G due to gravity of an electron located just above the surface of a white dwarf star. Note that the mass of the electron is 9.11 x 10^(-31) kg, the radius of the...
-
A wet bar of soap slides freely down a ramp 9.0 m long inclined at 6.09 Part A How long does it take to reach the bottom? Assume = 0.070 Express your answer using two significant figures. ? 5

Study smarter with the SolutionInn App


