From the outcome of the transformation shown in Fig. P6.48, indicate whether the levorotatory enantiomer of the
Question:
From the outcome of the transformation shown in Fig. P6.48, indicate whether the levorotatory enantiomer of the product has the R or S configuration. Draw a structure of the product that shows its absolute configuration.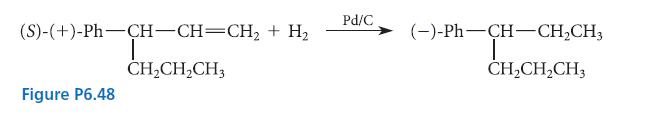
Transcribed Image Text:
(S)-(+)-Ph-CH-CH=CH₂ + H₂ I CH₂CH₂CH3 Figure P6.48 Pd/C (-)-Ph—CH-CH,CH, T CH₂CH₂CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
First draw the Senatiomer of the starting material using line ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Studies have shown that one stereoisomeric certain types of neurodegenerative disorders, system, as illustrated in structure B, and that form of compound A is an effective agent against Recognize...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The auditors report that follows was drafted by a staff accountant of Williams & Co., CPAs, at the completion of the audit of the financial statements of Lenz Corporation (nonpublic company) for the...
-
Assuming 10% minimum required reserve ratio, 5% excess reserve ratio, and people holding 10% of cash received at home (not deposited in the banks), the money multiplier is equal to Select one: a....
-
Suppose a defendant goes to a bar, gets blind drunk, and then goes out and robs a store. Is his intoxication a defense to the robbery?
-
How does a simplified employee pension plan differ from a Keogh plan? From a qualified pension plan?
-
The plaintiffs, Lee and Yoon, were the parents of H.L., a South Korean citizen who attended high school in Idaho as part of an exchange program. With permission from both plaintiffs and his host...
-
Financial information for Gallup Inc. is presented below. Instructions Prepare a schedule showing a horizontal analysis for 2014 using 2013 as the baseyear. Current assets Plant assets (net) Current...
-
1. Consider the recursive method whose definition appears below. public static String mysteryString (String s) { if (s.length()==1) else return s; } return s.substring (s.length()-1) + mysteryString...
-
(a) Explain why an optically inactive product is obtained when (2)-3-methyl-1-pentene undergoes catalytic hydrogenation. (b) What is the absolute configuration of (1)-3-methylhexane if catalytic...
-
(a) Give the stereochemical relationship (enantiomers, diastereomers, or the same molecule) between each pair of compounds in the set shown in Figure P6.46. Assume that internal rotation is rapid....
-
Show the products of these reaction and indicate which ismajor: N(CH3)3 N(CH3)3 a) b) d) c) CH3 .
-
Answer the following question: The SWOT analysis compares internal strengths and weaknesses with external opportunities and threats so that management identifies areas of concern and opportunity both...
-
Assume Phipps Chips, a fictious snack company, makes artisan potato chips in a variety of flavors. The company sells 12-ounce bags of a variety of flavors in grocery store chains such as Whole Foods...
-
With the growing complexity of managing worldwide supply chains, coupled with significant pressures to reduce cost at the firm level, supply managers must learn to manage the inherent risks of global...
-
CVS Case Discussion In 2002, CVS assembled a team of operations executives and managers for its Pharmacy Service Initiative (PSI). Although CVS was one of America's largest retail drugstores,...
-
Imagine you are attending a conference on psychotherapy. A therapist presents findings about a new form of therapy that has been developed and tested in a small sample of people. Patients' scores on...
-
Suppose a firm has only three possible plant-size options, represented by the ATC curves shown in the accompanying figure. What plant size will the firm choose in producing? (a) 50, (b) 130, (c) 160,...
-
Clark, PA, has been engaged to perform the audit of Kent Ltd.s financial statements for the current year. Clark is about to commence auditing Kents employee pension expense. Her preliminary enquiries...
-
An electrostatic potential map of (trifluoromethyl) benzene, C6H5CF3, is shown. Would you expect (trifluoromethyl) benzene to be more reactive or less reactive than toluene toward electrophilic...
-
Acetanilide is less reactive than aniline toward electrophilic substitution.Explain. c%3D0 Acetanilide CH
-
Draw resonance structures for the intermediates from reaction of an electrophile at the ortho, Meta and Para positions of nitrobenzene. Which intermediates are most stable?
-
Suppose that you have a sentineled, doubly-linked list implementation with the following methods: append_element (val). insert_element_at (val, index) remove_element_at (index) get_element_at (index)...
-
Consider the following algorithms. int add-them (int n, int A[]) { index i,j,k; j=0; for (i 1; in; i++) j=j+A: k=1; for (i=1;i; i++) k=k+k; return j+k; } int any equal (int n, int A[ ][ ]) { index...
-
1. Show that if the DES key K encrypts M to C, then K' encrypts M' to C'. That is: DESK (P) = C then DESK (P') C' 2. Show that AES decryption is indeed the inverse of the AES encryption. 3. Suppose...

Study smarter with the SolutionInn App


