Show the products of these reaction and indicate which ismajor: N(CH3)3 N(CH3)3 a) b) d) c)
Question:
Show the products of these reaction and indicate which ismajor:
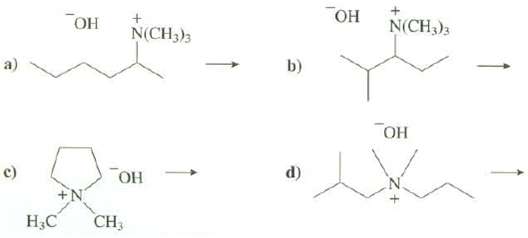
Transcribed Image Text:
Он Он N(CH3)3 N(CH3)3 a) b) Он d) c) ОН CH3 Н.С
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 78% (14 reviews)
Most E2 reactions follow Zaitsevs rule However the reaction kno...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Show the products of these reactions. (Remember that acid-base reactions are usually much faster than nucleophilic substitution reactions.) a) C1-CHCHCHCOH b) Br OH NH3 + OH
-
Show the products of these eliminationreactions: CH3 ELOH + NaOCH,CH3 a) "CI Br ELOH + NaOCH,CH; b) "CH,CH
-
Show the products of these reactions: Br DMSO + CH,CO, a) CI acetone + CH,CH,CO, b) CO DMF c) CH,CH CH,CH,Br +
-
Compensation survey data collected in July 2018 shows the average annual salary for Accountant II is $62,300.00 (rounded). With an aging rate of +2.3%, calculate the market value for an Accountant II...
-
Sugary Drink Ltd produces a soft drink called Fizz Cola, in two-litre bottles. Production takes place in two departments: Mixing and Packing. The manufacturing costs for each department for July...
-
Switching to an electronic bill-paying system would require a large capital expenditure for new computers and computer software. How could analyzing the companys income statement help you justify...
-
A normal shock occurs in a perfect gas. Sketch a Temperature - Entropy \((T-S)\) diagram of the process and show the following: Static and stagnation pressure and temperature before and after the...
-
The accounts of Polaris Company provided the following 20X5 information at 31 December 20X5: Accounts receivable ...............$ 1,899,000 (dr.) Allowance for sales discounts ...........23,500 (cr.)...
-
Let f(u) = u and g(x) = u = 3x6 +4. Find (fog)'(1). (fog) (1)= (Type an exact answer.)
-
Carmen Dahlen opened a secretarial school called Star office Training. a. Dahlen contributed the following assets to the business: Cash ........$5,700 Computers ....... 5,000 Office Equipments ......
-
The reaction of 2-bromobutane with ethoxide ion in ethanol gives 81% of a mixture of (Z)- and (E)-2-butene. Explain which stereo isomer you expect to predominate in this mixture.
-
Show the major products of these eliminationreactions: CI + NaOCH,CH3 Ph + NaOCH,CH, ELOH ELOH a) Ph Br d) Ph + NaOH CH;OH c) N(CH3)3 OTs
-
The TimpRiders LP has operated a motorcycle dealership for a number of years. Lance is the limited partner, Francesca is the general partner, and they share capital and profits equally. Francesca...
-
Of all 1,672,395 members of the high school class of 2014 who took the SAT (Scholastic Aptitude Test), 793,986 were minority students. Give the relevant proportion using correct notation.
-
The Dallas Development Corporation is considering the purchase of an apartment project for $\$ 100,000$. They estimate that they will receive $\$ 15,000$ at the end of each year for the next 10...
-
A Sampling Distribution for Average Salary of NFL Players Use StatKey or other technology to generate a sampling distribution of sample means using a sample of size \(n=5\) from the YearlySalary...
-
Raster Imaging Company has a new management team that has developed an operating plan to improve upon last year's ROE. The new plan would place the debt ratio at 55 percent, which will result in...
-
An ammeter that has internal resistance \(R_{\mathrm{am}}=0.504 \Omega\) is designed to measure a maximum current of \(I_{\max }=\) \(100 \mathrm{~mA}\). You want to use this ammeter to measure the...
-
For two inputs \(X_{1}\) and \(X_{2}\) and output \(Y\), a constant elasticity of substitution (CES) production function is given by where \(\alpha>0\) is an efficiency parameter, \(\eta>0\) is a...
-
Recall that Chapter 8 described the binary search algorithm for finding a particular entry in an ordered list. The idea behind binary search is to begin looking in the exact center of the list. If...
-
Solve each system by graphing. y=-3 5 y=-=x+2 2 7 3 --2-1 n y 2 3 LA x
-
Methyl-2-pentanone and 3-methylpentanal are isomers. Explain how you could tell them apart, both by mass spectrometry and by infraredspectroscopy. H. 4-Methyl-2-pentanone 3-Methylpentanal
-
Grignard reagents undergo a general and very useful reaction with ketones. Methyl magnesium bromide, for example, reacts with Cyclohexanone to yield a product with the formula C 7 H 14 O. What is the...
-
Ketones undergo a reduction when treated with sodium borohydride, NaBH 4 . What is the structure of the compound produced by reaction of 2-butanone with NaBH4 if it has an IR absorption at 3400 cm ?1...
-
003 The minimal polynomial associated with the matrix 102 01 1 is
-
Question 1: How do strategy and tactical action relate to each other in an organization? Question 2: How can you improve the implementation of strategy in this business organization?...
-
Larkin Corporation sells products for $125 each that have variable costs of $50 per unit. Larkin's annual fixed costs are $430,000. (10 points Total) a. What is Larkin's contribution margin per unit?...

Study smarter with the SolutionInn App


