Give the major product formed when each of the following amines is treated exhaustively with methyl iodide
Question:
Give the major product formed when each of the following amines is treated exhaustively with methyl iodide and then heated with Ag2O. Explain your reasoning.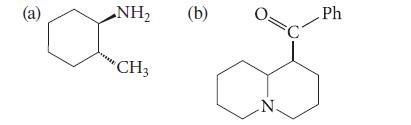
Transcribed Image Text:
(a) NH₂ CH3 (b) N. C Ph
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a The product of the exhaustive methylation followed by heatin...View the full answer

Answered By

Ehsan Mahmood
I’ve earned Masters Degree in Business Studies and specialized in Accounts & Finance. Couple with this, I have earned BS Sociology from renowned institute of Pakistan. Moreover, I have humongous teaching experience at Graduate and Post-graduate level to Business and humanities students along with more than 7 years of teaching experience to my foreign students Online. I’m also professional writer and write for numerous academic journals pertaining to educational institutes periodically.
4.90+
248+ Reviews
287+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Given the major product formed when each of the following amines is treated exhaustively with methyI iodide and then heated with Ag2O.Explain your reasoning. Ph N.
-
Give the major product formed when each of the following alcohols is heated in the presence of H2SO4; a. b. c. d. e. f. CH3CH2CH2CH2CH2OH CH CH3CH2C CHCH OH CH3 CHCH2CH3 OH CH,OH CH3 CH,CH2CH CCH OH...
-
Draw the major product(s) that are expected when each of the following amines is treated with excess methyl iodide and then heated in the presence of aqueous silver oxide. (a) (b) NH2 NH2
-
Your friend has just started a retail clothing store in kamloops. She will be purchasing inventory to make her own clothing (One Style) and she will also be buying ready to sell iteams. She has come...
-
Identify the three types of ownership structures and discuss the advantages and disadvantages of each.
-
How do management reports produced by a public accounting firms engagement management system for the current year engagement assist them in bidding on the job in subsequent years? In justifying to...
-
Using only the factor formulas given in Table 2.6, derive Equation 7.6 starting with Equation 7.3. TABLE 2.6 Summary of Discrete Compounding Interest Factors. To Find Given Factor Symbol Name P F...
-
Horace Greeley Corporation was organized in 2013 and began operations at the beginning of 2014. The company is involved in interior design consulting services. The following costs were incurred prior...
-
3. (a) Find the binary representation of /3. (b) By noting that -9.6= (-1) 23 1.2. x Use the result in (a), represent -9.6 in IEEE single precision format through rounding. (c) Represent the result...
-
Provide a reaction mechanism for the reaction shown in Eq. 23.36. NH 5- + 3 Br Br NH Br Br + 3HBr (23.36)
-
Suggest two syntheses of N-ethylcyclohexanamine by reductive amination.
-
Procter & Gamble will pay an annual dividend of $0.65 one year from now. Analysts expect this dividend to grow at 12% per year thereafter until the fifth year. After then, growth will level off at 2%...
-
Your company has several running projects that were financed with various types of loans. It would like to start one more project, and finance the purchase of PROFIT the production equipment when the...
-
Your application is using ASG, and an ALB distributes traffic to the ASG. You want to add a scaling policy that keeps the average aggregate CPU utilization of the ASG to be 8 0 percent. The capacity...
-
Data for Questions 1 and 2 A reservoir has a drainage radius of 1500 ft, thickness of 100 ft, horizontal permeability of 20 md and a vertical permeability of 2 md. The oil viscosity is 2.5 cp and the...
-
Determine expressions for the mean residence time given "outward" diffusional release into a perfect sink from a) A cylindrical monolithic device of radius a, with ends capped. b) A spherical device...
-
You work as a financial analyst for the CFO of a big company, you have been asked to consider the following cash flows (Millions) from two mutually exclisive capital budgeting projects. If the firm's...
-
What does gradual refinement mean in the context of SDLC?
-
Halley's comet travels in an ellipti- cal orbit with a = 17.95 and b = 4.44 and passes by Earth roughly every 76 years. Note that each unit represents one astronomical unit, or 93 million miles. The...
-
In the previous problem and the associated molecular model at the book's website, you considered the role of HOMOs and LUMOs in an SN2 reaction. (a) What is the LUMO in an SN1 reaction and in what...
-
SN2 reactions that involve breaking a bond to a chirality center can be used to relate configurations of molecules because the stereochemistry of the reaction is known. (a) Illustrate how this is...
-
Keeping in mind that carbocations have a trigonal planar structure, (a) Write a structure for the carbocation intermediate (b) Write structures for the alcohol (or alcohols) that you would expect...
-
For each of the following independent cases (A through E), compute the missing values in the table: Prime Conversion Case Cost Cost Direct Materials Direct Labor Manufacturing Overhead Total...
-
Most businesses sell several products at varying prices. The products often have different unit variable costs. Thus, the total profit and the breakeven point depend on the proportions in which the...
-
Your friend, Suzie Chang, has designed a new type of outdoor toy that helps children learn basic concepts such as colors, numbers, and shapes. Suzie's product will target two groups: day care centers...

Study smarter with the SolutionInn App


